Chapter: Basic & Clinical Pharmacology : Agents Used in Anemias; Hematopoietic Growth Factors
Erythropoietin - Hematopoietic Growth Factors
ERYTHROPOIETIN
Chemistry & Pharmacokinetics
Erythropoietin, a
34–39 kDa glycoprotein, was the first human hematopoietic growth factor to be
isolated. It was originally puri-fied from the urine of patients with severe
anemia. Recombinant human erythropoietin (rHuEPO, epoetin alfa) is produced in
a mammalian cell expression system. After intravenous administra-tion,
erythropoietin has a serum half-life of 4–13 hours in patients with chronic
renal failure. It is not cleared by dialysis. It is mea-sured in international
units (IU). Darbepoetin alfa is a modified form of erythropoietin that is more
heavily glycosylated as a result of changes in amino acids. Darbepoetin alfa
has a twofold to threefold longer half-life than epoetin alfa. Methoxy
polyethylene glycol-epoetin beta is an isoform of erythropoietin covalently
attached to a long polyethylene glycol polymer. This long-lived recombinant
product is administered as a single intravenous or subcutaneous dose at 2-week
or monthly intervals, whereas epoe-tin alfa is generally administered three
times a week and darbepo-etin is administered weekly.
Pharmacodynamics
Erythropoietin
stimulates erythroid proliferation and differentia-tion by interacting with
erythropoietin receptors on red cell pro-genitors. The erythropoietin receptor
is a member of the JAK/ STAT superfamily of cytokine receptors that use protein
phospho-rylation and transcription factor activation to regulate cellular
function . Erythropoietin also induces release of reticulocytes from the bone
marrow. Endogenous erythropoietin is primarily produced in the kidney. In
response to tissue hypoxia, more erythropoietin is produced through an
increased rate of transcription of the erythropoietin gene. This results in
correction of the anemia, provided that the bone marrow response is not
impaired by red cell nutritional deficiency (especially iron defi-ciency),
primary bone marrow disorders , or bone marrow suppression from drugs or
chronic diseases.Normally, an inverse relationship exists between the
hematocrit or hemoglobin level and the serum erythropoietin level. Nonanemic
individuals have serum erythropoietin levels of less than 20 IU/L. As the
hematocrit and hemoglobin levels fall and anemia becomes more severe, the serum
erythropoietin level rises exponentially. Patients with moderately severe anemia
usually have erythropoie-tin levels in the 100–500 IU/L range, and patients
with severe anemia may have levels of thousands of IU/L. The most important
exception to this inverse relationship is in the anemia of chronic renal
failure. In patients with renal disease, erythropoietin levels are usually low
because the kidneys cannot produce the growth factor. These are the patients
most likely to respond to treatment with exogenous erythropoietin. In most
primary bone marrow disorders (aplastic anemia, leukemias, myeloproliferative
and myelodysplastic disorders, etc) and most nutritional and second-ary
anemias, endogenous erythropoietin levels are high, so there is less likelihood
of a response to exogenous erythropoietin .
Clinical Pharmacology
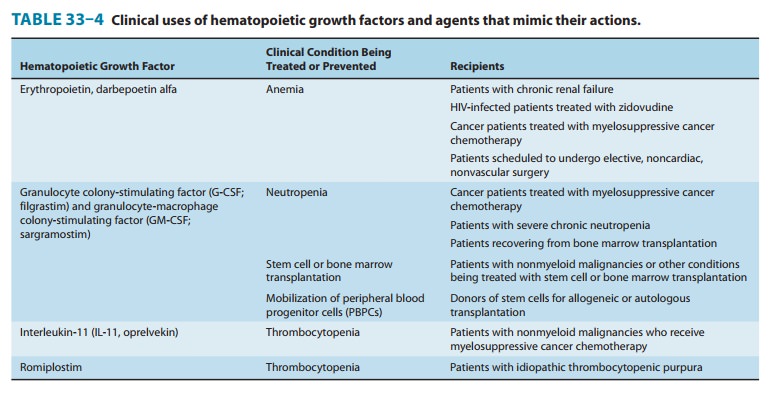
The availability of erythropoiesis-stimulating agents (ESAs) has had a significant positive impact for patients with several types of anemia (Table 33–4).
The ESAs consistently improve the hematocrit
and hemoglobin level, often eliminate the need for transfusions, and reliably
improve quality of life indices. The ESAs are used routinely in patients with
anemia secondary to chronic kidney disease. In patients treated with an ESA, an
increase in reticulocyte count is usually observed in about 10 days and an
increase in hematocrit and hemoglobin levels in 2–6 weeks. Dosages of ESAs are
adjusted to maintain a target hemoglobin up to, but not exceeding, 10–12 g/dL.
To support the increased erythropoiesis, nearly all patients with chronic
kidney disease require oral or parenteral iron supplementation. Folate
supplementation may also be necessary in some patients.
In
selected patients, erythropoietin is also used to reduce the need for red blood
cell transfusion in patients undergoing myelo-suppressive cancer chemotherapy
who have a hemoglobin level <10 mg/dL, and for selected patients with
low-risk myelodysplas-tic syndromes and anemia requiring red blood cell
transfusion. Patients who have disproportionately low serum erythropoietin
levels for their degree of anemia are most likely to respond to treat-ment.
Patients with endogenous erythropoietin levels of less than 100 IU/L have the
best chance of response, although patients with erythropoietin levels between
100 and 500 IU/L respond occa-sionally. Methoxy polyethylene glycol-epoetin
beta should not be used for treatment of anemia caused by cancer chemotherapy
because a clinical trial found significantly more deaths among patients
receiving this form of erythropoietin.hemoglobin levels are <10 g/dL) and
with the lowest dose needed to avoid transfusion.
Allergic
reactions to ESAs have been infrequent. There have been a small number of cases
of pure red cell aplasia (PRCA) accompanied by neutralizing antibodies to
erythropoietin. PRCA was most commonly seen in dialysis patients treated
subcutane-ously for a long period with a particular form of epoetin alfa (Eprex
with a polysorbate 80 stabilizer rather than human serum albumin) that is not
available in the United States. After regula-tory agencies required that Eprex
be administered intravenously rather than subcutaneously, the rate of
ESA-associated PRCA diminished. However, rare cases have still been seen with
all ESAs administered subcutaneously for long periods to patients with chronic
kidney disease.
Related Topics