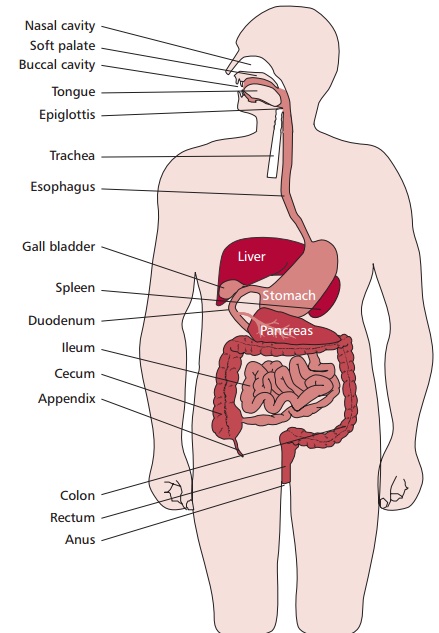
Chapter: Biology of Disease: Disorders of the Gastrointestinal Tract, Pancreas, Liver and Gall Bladder
Digestion: Mouth, Stomach, Small Intestine
DIGESTION
Digestion is the hydrolytic breakdown of nutrient macromolecules and compound lipids to smaller products that can be absorbed. The hydrolytic reactions are catalyzed by a variety of enzymes: proteases that digest pro-teins; carbohydrases that digest carbohydrates; lipases that catalyze the hydrolysis of lipids and nucleases that degrade DNA and RNA. Digestion occurs in the mouth, to a small extent, stomach and small intestine, and most absorption of nutrients occurs in the small intestine and that of water in the large intestine.
MOUTH
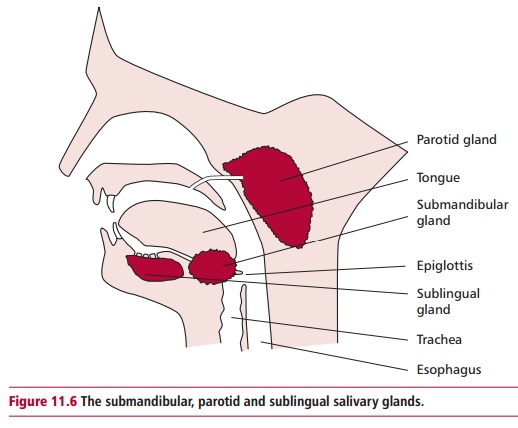
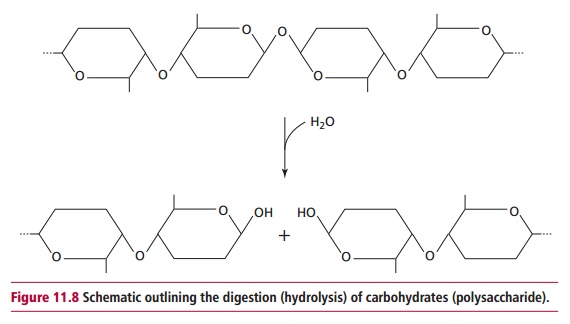
In the mouth, teeth break the food into smaller portions increasing the surface area upon which digestive enzymes can act. Three pairs of salivary glands, the submandibular located under the jaws, the sublingual located under the tongue and the parotid situated in front of the ears (Figure 11.6), secrete saliva into the mouth. The saliva contains amylase (Figure 11.7), water and mucus. The water helps to dissolve nutrients, while the mucus acts as a lubricant to aid swallowing and lubricates passage of food through the GIT. Chewing and the actions of the tongue mix the food with the saliva. Salivary amylase begins the digestion of carbohydrates (Figure 11.8) although the digestion of carbo-hydrates in the mouth is minimal since the food is not retained there for any length of time. Lingual lipase is secreted by lingual serous glands. Again, little digestion occurs in the mouth but it has been suggested that the fatty acids
In swallowing, the soft palate is elevated, which seals off the nasal cavity and pushes against the back wall of the pharynx. This, in turn, triggers an auto-matic reflex action in muscles that raise the larynx, pushing its opening, the glottis, against a flap of tissue called the epiglottis. These actions prevent food entering the trachea of the respiratory system and ensure food is expelled from the mouth into the esophagus. Rhythmic waves of contraction of smooth muscle tissue in the walls of the GIT, calledperistalsis, forces food along the GIT. Peristalsis in the esophagus ensures that food is propelled to the stomach even if the person is upside down.
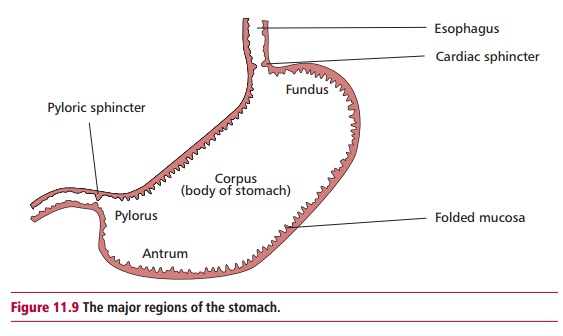
STOMACH
The stomach is a sac-like region of the GIT (Figure 11.1 (A) and (B)). Its inner surface is highly folded (Figure 11.9) allowing it to expand up to eightfold from its empty volume of about 50 cm3 following a meal. Muscular activities of the stomach wall mix the food with gastric juice secreted by gastric glands located in the mucosal lining of the stomach. Gastric glands produce about 1 to 2 dm3
of gastric juice per day from three types of secretory cells. Parietal or oxyntic cells produce hydrochloric acid (HCl), peptic or chief cells secrete pepsino-gen, the precursor of pepsin, and gastric lipase, and mucous cells that secrete mucus that protects the mucosa lining the stomach from the corrosive action of the HCl.
The activity of salivary amylase continues within the bolus and is possibly only inactivated when the bolus contents are completely mixed with the acidic gastric juice. The pH optimum of lingual lipase is between 3.5 and 6.0 and is activated in the upper portion of the stomach. Gastric lipase is secreted by the chief cells of the stomach and also has an acid optimum. About 10 to 30% of dietary fat may be hydrolyzed in the stomach. However, lipase activity in the stomach is highest against triacylglycerols with short or medium length fatty acid residues, which are found in milk, and so may be of most importance in newborn infants.

Pepsinogen is a weakly-active protease. It has an acid optimum pH, hence the need for HCl secretion in the stomach. In these conditions, about pH 1 to 2, protein molecules in the food are denatured making them more susceptible to digestion. Also, at this pH, pepsinogen molecules act on one another at specific sites to produce the fully active protease, pepsin (Figure 11.10), that begins the digestion of the denatured proteins to form shorter polypeptides and peptides. The denaturation and digestion of proteins reduces the chance of their absorption and prevents them being immunogenic . The watery mixture of gastric juice and partially digested food is called chyme.
The acid also activates intrinsic factor (IF), a glycoprotein secreted by the pari-etal cells that is needed for absorption of vitamin B12 . Vitamin B12 is released from dietary proteins by the action of pepsin and binds to one of two binding proteins present in gastric juice whose affinity for vitamin B12 is increased in acid conditions and is greater than that of IF. When chyme enters the small intestine, proteases from pancreatic juice break down the binding proteins and vitamin B12 becomes bound to IF. The mucosal lining in the ileum has receptors for IF which bind the IF–vitamin B12 complex, so that vitamin B12 is absorbed and enters the portal blood. Given that the absorption of vita-min B12 is dependent upon IF, any condition that decreases the secretion of IF, for example atrophic gastritis, interferes with digestion of the binding proteins, for example pancreatic exocrine insufficiency, or decreases the binding and internalization of the IF–vitamin B12 complex, such as some diseases that affect the ileum, can cause pernicious anemia .
The acid environment of the stomach kills most of the bacteria that are ingested with the food but some survive and enter the small intestine in the chyme. The pyloric sphincter controls the passage of chyme from the stom-ach to the small intestine allowing only relatively small amounts of chyme through at any one time. This control is necessary because the digestive activ-ity within the small intestine is time-consuming and because its capacity is limited. The activities of the stomach and small intestine are coordinated by the nervous and endocrine systems.
SMALL INTESTINE
The final stages of digestion and the absorption of its products occur in the small intestine (Figure 11.1 (A)). Contractions in the small intestine help to break food up, mix it with digestive juices and propel it towards the colon. The small intestine is about 23 feet (7 m) long in an adult. It is divided into three sections: the duodenum constitutes the first 250 mm, followed by the jejunum then the ileum. Digestion occurs primarily in the duodenum and jejunum and absorption in the ileum.
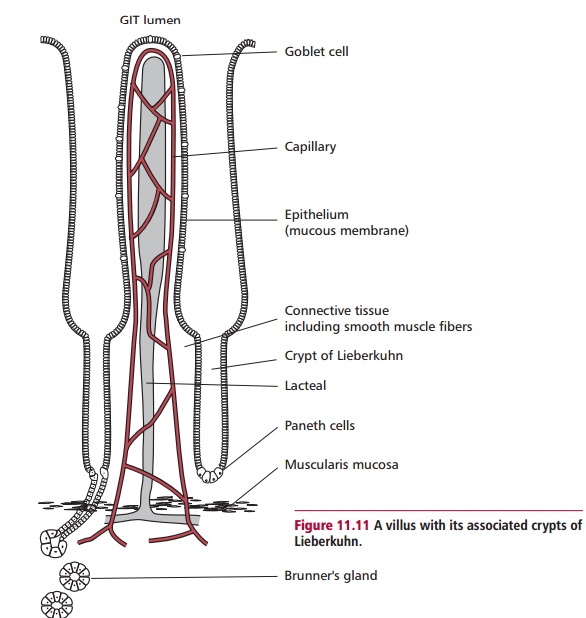
The walls of the duodenum contain goblet cells that secrete mucus that protects it against damage by acidic chyme. Crypts of Lieberkuhn are pits within the wall of the small intestine with secretory cells that release intes-tinal juice and Paneth cells that secrete lysozyme. Intestinal juice is largely water with mucus and buffered to a pH of about 7.6. Along with pancreatic juice (see below), it neutralizes chyme and provides a liquid medium that aids absorption of nutrients. Lysozyme is an antibacterial enzyme. The epi-thelial lining consists of cells called enterocytes organized into small pro-jections into the lumen called villi (Figure 11.11). In turn, each enterocyte
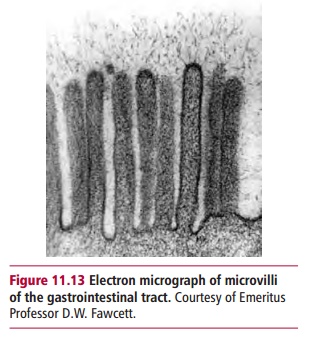
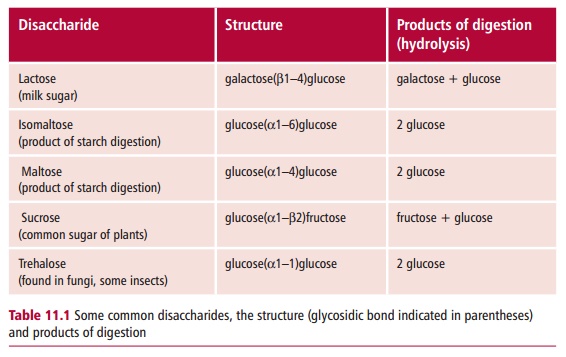
has its apical plasma membrane surface, which is the side facing the lumen of the GIT, arranged into microscopic extensions called microvilli forming a brush border that coats the villi (Figure 11.13). The opposing face of the enterocyte is called the basolateral membrane. The presence of villi and microvilli means that the interior surface area of the small intestine is enor-mous, estimated at 300 m2 in humans. Hydrolytic enzymes that are integral proteins of the plasma membranes forming the microvilli catalyze further digestive reactions. For example: lactose is hydrolyzed to glucose and galac-tose in a reaction catalyzed by lactase; sucrose to glucose and fructose by sucrase; and the disaccharides maltose and isomaltose, produced by the action of amylase on starch, are hydrolyzed to glucose by maltase and iso-maltase activities (Table 11.1).
Pancreatic juice is produced by the pancreas and contains water, alkaline salts that give it a pH of 7.8 to 8.0, enzymes and zymogens and
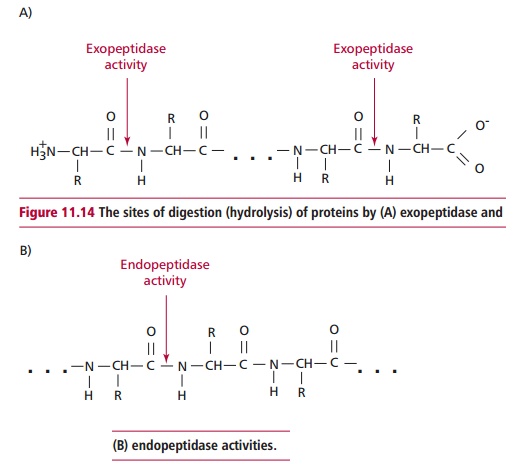
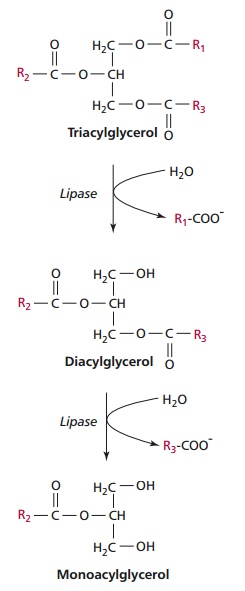
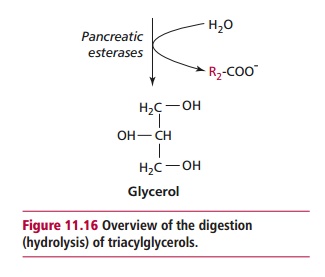
Proteolytic enzymes can be divided into exopeptidases and endopeptidases. The exopeptidases are aminopeptidases and carboboxypeptidases that cata-lytically remove amino acids from the ends of proteins and peptides (Figure11.14 (A)). The pancreatic proteases, elastase, trypsin and chymotrypsin, areendoproteases (Figure 11.14 (B)) and hydrolyze proteins and peptides at dif-ferent peptide bonds throughout the molecules to produce peptides and amino acids. The actions of endopeptidases increase the number of protein ends, effectively increasing the concentration of substrates for the exopepti-dases. Pancreatic amylase continues the digestion of polysaccharides to pro-duce disaccharides. Pancreatic lipase (Figure 11.15) hydrolyzes lipids to fatty acids and 2-monoacylglycerols but requires the presence of colipase, another protein secreted by the pancreas, for full activity. Bile released from the gall bladder enters the intestine through the bile duct and emulsifies lipids into droplets of about 1 Lm diameter, which greatly increases the surface area available for lipase activity. Pancreatic esterase activity can complete the digestion to glycerol and free fatty acids (Figure 11.16).
The activities of many of the enzymes involved in digestion are summarized
Related Topics