Chapter: Modern Medical Toxicology: Neurotoxic Poisons: Drugs Used in Psychiatry
Cyclic Antidepressants
Cyclic Antidepressants
·
Tricyclic antidepressants possess a
3-ring molecular structure. Examples include amitriptyline, clomipramine,
desipramine, dibenzepin, doxepin, dothiepin, imipramine, lofepramine,
nortriptyline, protriptyline and trimipramine.
Uses
â– â– Tricyclic
antidepressants are used to treat a wide range of disorders such as depression,
panic disorder, social phobia, bulimia, narcolepsy, attention deficit disorder,
obsessive compulsive disorder, childhood enuresis, and chronic pain syndromes.
Toxicokinetics
·
All cyclic antidepressants are rapidly absorbed from the GI
tract and have large volumes of distribution (10 to 50 L/kg). Most of them bind
to plasma protein alpha1-glycoprotein with varying affinity.
·
They are all highly lipophilic, sparingly water soluble, and
substantially metabolised by first-pass in the liver. The metabolites retain
significant pharmacologic activity until hydroxylation occurs by microsomal
enzyme system.
·
The half-lives of these compounds are highly variable (4 hrs
to 93 hrs).
·
Approximately 30% of the absorbed dose is eliminated by
gastric and biliary secretion, while renal clearance accounts for 3 to 10% of
the parent compound. It is however important to remember that cyclic
antidepressants taken in large quantities (overdose) exhibit significantly
altered toxicokinetics.
o
Absorption may be delayed by inhibition of gastric emptying
and peristalsis.
o
Enterohepatic recirculation delays final elimination of a
large amount of the drug.
o
Enzymes responsible for hydroxylation can become saturated.
o
The amount of drug unbound to plasma proteins may increase
(because of acidaemia).
Mode of Action
·
Cyclic antidepressants, notably the tricyclics, are
structur-ally similar to the phenothiazines with similar anticho-linergic,
adrenergic, and alpha-blocking properties of the phenothiazines.
·
Following absorption, these agents are extensively bound to
plasma proteins and also bind to tissue and cellular sites, including the
mitochondria.
·
Cyclic antidepressants act by inhibiting voltage-gated sodium
channels in myocardial cells, blocking of H1, H2, and D2
receptors, as well as muscarinic receptors, inhibiting alpha-adrenergic
receptors, interacting with GABA recep- tors, and inhibiting the transport and
reuptake of biogenic amines at nerve terminals.
·
The toxicity of cyclic antidepressants is mainly due to
effects on myocardium, CNS, and peripheral vasculature. There is prolongation
of action potential duration in most myocardial cells, decreased peripheral
vascular resistance, and induc- tion of anticholinergic effects. Convulsions
resulting from overdose are caused by complicated interactions within the brain
due to altered concentrations of GABA, dopamine, noradrenaline and
acetylcholine.
Adverse Effects
·
Postural hypotension, cardiac
arrhythmias, vertigo, weak- ness, tremor, confusion, weight gain,
agranulocytosis and thrombocytopenia.
·
Anticholinergic effects are common:
tachycardia, hyper- tension, mydriasis, dry and flushed skin, visual blurring,
decreased GI motility (constipation), urinary retention, and delirium with
hallucinations and convulsions.
·
Abrupt withdrawal of a chronically
administered cyclic antidepressant can cause a cholinergic rebound syndrome: anorexia, nausea, vomiting,
diarrhoea, sweating, myalgia, headache, fatigue, anxiety, insomnia, mania,
akithisia or Parkinsonism.
Drug Interactions
·
Cyclic antidepressants potentiate
the sedative effect of alcohol, and the hypertensive effect of
sympathomimetics. They aggravate the anticholinergic effect of antiparkin-They
aggravate the anticholinergic effect of antiparkin- pyrexia with convulsions
and coma when combined with MAOIs.
·
The effect of antihypertensive drugs
is reduced.
·
Serotonin reuptake inhibitors
inhibit cytochrome P450-2D6 and can cause elevations in serum tricyclic
antidepressant levels.
·
While tricyclic antidepressants and
MAOIs have been used concomitantly to treat severe depression, the combination
has occasionally been associated with the development of serotonin syndrome.
Overdose with the combination of tricyclic antidepressants and MAOI appears to
cause severe effects and has a high fatality rate.
Clinical (Toxic) Features
·
Overdose results in seizures
(especially common with maprotiline and amoxapine), which are generally brief in duration,
tachycardia,* hypotension, agitation, hallu-cinations, confusion including
anticholinergic delirium, hyperthermia, ataxia, urinary retention, and coma.
Coma is usually short-lived, and most patients waken within 24 hours.
·
Anticholinergic effects (mydriasis, tachycardia, urinary
retention, decreased gastrointestinal motility) are common, but may be masked
in severe overdose.
·
Miosis may be present in deeply comatose patients. Nystagmus
may occur.
·
Rhabdomyolysis and renal failure may result from prolonged
seizures or coma.
·
Significant metabolic acidosis may develop in patients with
prolonged seizures or hypotension.
·
Neuroleptic malignant syndrome (NMS) has been reported.
·
Respiratory depression is common with significant over-doses
and may develop rapidly. Adult respiratory distress syndrome may occur after
severe overdose.
·
Cardiovascular toxicity results in myocardial depression,
ventricular tachycardia, and fibrillation. A variety of ECG findings have been
described (Table 19.4). Severe
cardiac toxicity generally develops within six hours, although ECG changes may
persist beyond 48 hours.

· Myocardial infarction has been reported following over-dose and with therapeutic use. The duration of coma with cyclic antidepressant overdose is generally less than 6 to 12 hours. If this is prolonged beyond 24 hours, it indicates development of complica-tions or concomitant ingestion of CNS depressants.
·
Amitryptiline overdose may be associated with peripheral
neuropathy, polyradiculoneuropathy, and extrapyramidal manifestations.
·
Radiological evidence of pulmonary oedema is present in 10
to 15% of patients with cyclic antidepressant over-dose: diffuse bilateral
pulmonary infiltrates and other features characteristic of ARDS.
·
Uncommon manifestations include fulminant hepatic failure,
bowel ischaemia, and acute intestinal pseudo-obstruction. Pruritic erythematous
rash, vesicular erup-tion, blistering and skin discolouration have also been
reported.
·
Withdrawal syndromes may occur after discontinuation of
tricyclic antidepressants. Symptoms associated with tricyclic antidepressant
withdrawal may include nausea, diarrhoea, malaise, myalgias, headache,
rhinorrhoea, anxiety, agitation, mania, insomnia, nightmares, arrhyth-mias and
ventricular ectopy.
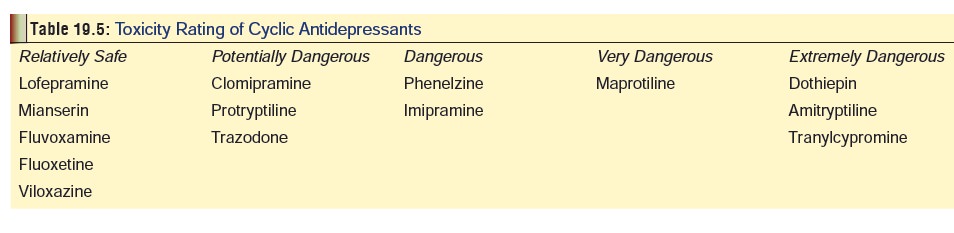
·
A suggested toxicity rating for various cyclic
antidepres-sants in overdose is outlined in Table 19.5.
Usual Fatal Dose
â– â– Death is not uncommon in severe cyclic antidepressant
overdose, and is more likely in the presence of the following factors:
·
Age > 30 years.
·
Serum drug level > 800 ng/ml
(2880 mmol/L).
·
Ingestion of amitryptiline.
·
Heart rate > 120.
·
QRS duration > 100 ms.
·
QRS axis > 90°.
·
Terminal 40-ms axis > 135°.
· QTc interval > 480 ms.
â– â– Serum drug level of more than 1000 ng/ml (10 to 20 mg/kg PO)
is usually fatal. The therapeutic range for most tricyclic antidepressants is
100 to 260 ng/ml.
â– â– Ten times the therapeutic daily dose of a cyclic antidepres-
sant is potentially fatal.
â– â– Fatal poisonings have occurred in children following the
ingestion of as little as 250 mg of imipramine or amoxapine.
Diagnosis
·
Monitor serum electrolytes, renal
and hepatic function in patients with significant toxicity.
·
Follow CPK levels in patients with
prolonged seizures or coma.
·
Serum tricyclic levels are useful in
initial assessment since they may serve to confirm a history of ingestion.
·
ECG changes (vide supra). Monitor serial ECGs and institute continuous cardiac
monitoring in all patients with suspected tricyclic overdose. ECG changes may
include sinus tachycardia, prolonged PR interval, widening of the QRS complex,
QTc prolongation, rightward shift in the axis of the terminal 40 milliseconds
of the QRS complex, Twave flattening or inversion, ST segment depression, right
bundle branch block, junctional rhythm and atrioventricular block.
·
Elevation of creatine kinase and
lactic acid dehydrogenase levels.
·
Chest X-ray to detect pulmonary
oedema.
Treatment
1.
Patients with ECG changes should be monitored in the ICU until the mental
status is baseline, the patient is asymptomatic, and the ECG has returned to
normal for 24 hours. Monitor cardiac rhythm and serial ECGs. Maximal limb-lead
QRS duration of 0.10 seconds or longer has been associated with an increased
incidence of seizures, while a QRS of 0.16 seconds or longer has been
associated with an increased incidence of ventricular dysrhythmias. A terminal
40 ms QRS axis of >120 degrees or an R wave in lead aVR of > 3 mm are
thought to be a more sensitive indicator of tricyclic antidepressant toxicity
than QRS interval, although they have not been correlated with outcome or
complica-tions.
2. Supportive measures:
·
Maintain airway; intubate if
indicated.
·
Monitor arterial blood gases.
·
Administer oxygen if necessary.
· Treat hypotension with IV crystalloids, inotropes (dopamine), vasopressors (noradrenaline), etc., as necessary. Intra-aortic balloons have been used success-fully when pressors have failed.
3. Reduce drug absorption:
·
Stomach wash (within the first 6
hours).
·
Activated charcoal (1 gm/kg).
4. Enhance drug elimination:
·
Multiple-dose activated charcoal.
·
Diuresis and haemodialysis are not
effective.
·
Haemoperfusion is not routinely
recommended, but has been used in patients with severe intoxication.
5. Treat convulsions:
·
Diazepam 0.1 mg/kg IV
·
Phenytoin 15 mg/kg IV infusion.
·
If seizures cannot be controlled
with diazepam or phenytoin, or recur, administer phenobarbitone. If
phenobarbitone is ineffective, consider paralysis and/ or barbiturate coma.
6. Treat arrhythmias:
·
Serum alkalinisation to a pH of 7.45 to 7.55 using
intra-venous boluses of sodium bicarbonate is recommended for patients with
arrhythmias or QRS widening.
·
Intubation and hyperventilation may be used as an adjunct to
sodium bicarbonate to achieve serum alka-linisation, with careful monitoring of
blood gases to avoid profound alkalaemia.
·
Conventional antiarrhythmics may also be necessary.
Quinidine, disopyramide, and procainamide are type 1a and are contraindicated,
as their effects on myocardial conduction are similar to that of the tricyclic
antidepres-sants.
·
Increased QRS duration may be the best indication of
severity of overdose and risk of serious complications, and should be treated
aggressively.
·
Sinus tachycardia—supportive measures only.
·
Supraventricular arrhythmias—alkalinise (to 7.40 – 7.45 pH);
synchronised cardioversion if alkalinisation is ineffective. 1 to 2 mEq/kg of
sodium bicarbonate is administered as needed to achieve a physiologic pH, or
slightly above (7.45 to 7.55). A pH greater than 7.60 or a pCO2 less than 20
mmHg is probably undesirable. Effective alkalinisation may not be achievable by
using intravenous continuous infusion of sodium bicarbonate with conventional
doses (2 ampoules per litre).
·
Ventricular tachycardia—alkalinise (to 7.40 – 7.45 pH);
lignocaine 1mg/kg IV, bolus, followed by infusion of 2 to 4 mg/min;
synchronised cardioversion if these measures are ineffective; isoprenaline
infusion 0.5 to 5.0 mcg/min and overdrive pacing for torsade de pointes.
·
Ventricular fibrillation—defibrillate; sodium bicarbo-nate 1
to 3 mmol/kg, and hyperventilation for achieving a pH of 7.45 – 7.50; 1: 1000
adrenaline, 0.5 to 1.0 mg IV; lignocaine 1 mg/kg IV bolus, followed by 2 to 4
mg/min infusion; beta blockers if these measures are ineffective.
·
Bradycardia or heart block—alkalinise to 7.40 to 7.45 pH;
isoprenaline; pacemaker.
·
Refractory cardiac arrest—basic and advance life support for
a minimum of 1 hour; alkalinise to 7.5 pH.
·
Hypertonic saline has been found to be useful in some cases.
·
Use of physostigmine in the setting of tricyclic
anti-depressant overdose is controversial and has been associated with the
development of seizures and fatal dysrhythmias. It is not recommended except to
treat life-threatening symptoms that have been unresponsive to other therapies.
·
Flumazenil is contraindicated even if benzodiazepines are
known coingestants; use of flumazenil in the setting of tricyclic
antidepressant overdose has been associated with the onset of seizures and
ventricular arrhythmias.
Related Topics