Chapter: Medical Physiology: Reproductive and Hormonal Functions of the Male (and Function of the Pineal Gland)
Control of Male Sexual Functions by Hormones from the Hypothalamus and Anterior Pituitary Gland
Control of Male Sexual Functions by Hormones from the Hypothalamus and Anterior Pituitary Gland
A major share of the control of sexual functions in both the male and the female begins with secretion of gonadotropin-releasing hormone (GnRH) by the hypothalamus (see Figure 80–10). This hormone in turn stimulates the anterior pituitary gland to secrete two other hormones called gonadotropic hormones: (1) luteinizing hormone (LH) and (2) follicle-stimulat-ing hormone (FSH). In turn, LH is the primary stimu-lus for the secretion of testosterone by the testes, and FSH mainly stimulates spermatogenesis.
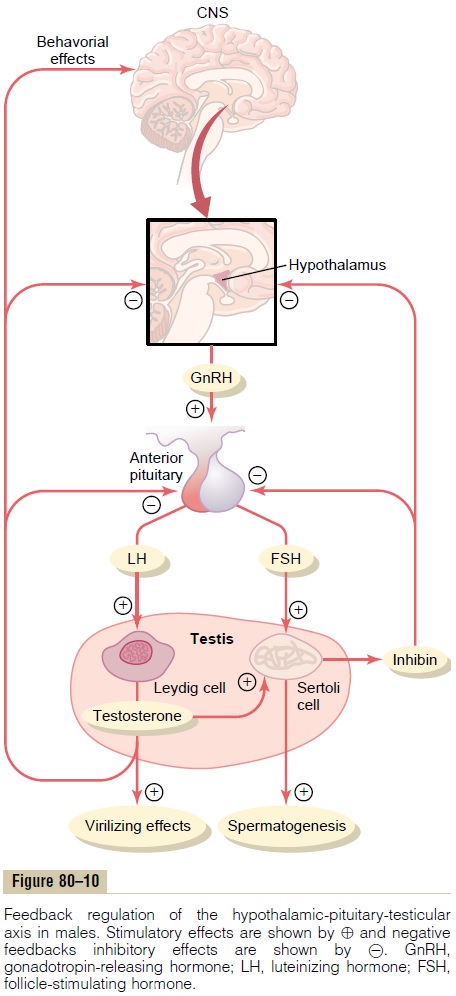
GnRH and Its Effect in Increasing the Secretion of LH and FSH GnRH is a 10-amino acid peptide secreted by neurons whose cell bodies are located in the arcuate nuclei ofthe hypothalamus. The endings of these neuronsterminate mainly in the median eminence of the hypothalamus, where they release GnRH into the hypothalamic-hypophysial portal vascular system. Then the GnRH is transported to the anterior pituitary gland in the hypophysial portal blood and stimulates the release of the two gonadotropins, LH and FSH.
GnRH is secreted intermittently a few minutes at a time once every 1 to 3 hours. The intensity of this hormone’s stimulus is determined in two ways: (1) by the frequency of these cycles of secretion and (2) by the quantity of GnRH released with each cycle.
The secretion of LH by the anterior pituitary gland is also cyclical, with LH following fairly faithfully the pulsatile release of GnRH. Conversely, FSH secretion increases and decreases only slightly with each fluctuation of GnRH secretion; instead, it changes more slowly over a period of many hours in response to longer-term changes in GnRH. Because of the much closer relation between GnRH secretion and LH secretion, GnRH is also widely known as LH-releasing hormone.
Gonadotropic Hormones: LH and FSH
Both of the gonadotropic hormones, LH and FSH, are secreted by the same cells, called gonadotropes, in the anterior pituitary gland. In the absence of GnRH secretion from the hypothalamus, the gonadotropes in the pituitary gland secrete almost no LH or FSH.
LH and FSH are glycoproteins. They exert their effects on their target tissues in the testes mainly by activating the cyclic adenosine monophosphate second messenger system, which in turn activates specificenzyme systems in the respective target cells.
Testosterone—Regulation of Its Production by LH. Testos-terone is secreted by the interstitial cells of Leydig inthe testes, but only when they are stimulated by LH from the anterior pituitary gland. Furthermore, the quantity of testosterone secreted increases approxi-mately in direct proportion to the amount of LH available.
Mature Leydig cells are normally found in a child’s testes for a few weeks after birth but then disappear until after the age of about 10 years. However, either injection of purified LH into a child at any age or secretion of LH at puberty causes testicular interstitial cells that look like fibroblasts to evolve into function-ing Leydig cells.
Inhibition of Anterior Pituitary Secretion of LH and FSH by Testosterone—Negative Feedback Control of Testosterone Secretion. The testosterone secreted by the testes inresponse to LH has the reciprocal effect of inhibiting anterior pituitary secretion of LH (see Figure 80–10). Most of this inhibition probably results from a direct effect of testosterone on the hypothalamus to decrease the secretion of GnRH. This in turn causes a corre-sponding decrease in secretion of both LH and FSH by the anterior pituitary, and the decrease in LH reduces the secretion of testosterone by the testes. Thus, whenever secretion of testosterone becomes too great, this automatic negative feedback effect, operat-ing through the hypothalamus and anterior pituitary gland, reduces the testosterone secretion back toward the desired operating level. Conversely, too little testosterone allows the hypothalamus to secrete large amounts of GnRH, with a corresponding increase in anterior pituitary LH and FSH secretion and conse-quent increase in testicular testosterone secretion.
Regulation of Spermatogenesis by FSH and Testosterone
FSH binds with specific FSH receptors attached to the Sertoli cells in the seminiferous tubules. This causes these cells to grow and secrete various spermatogenic substances. Simultaneously, testosterone (and dihy-drotestosterone) diffusing into the seminiferous tubules from the Leydig cells in the interstitial spaces also has a strong tropic effect on spermatogenesis. Thus, to initiate spermatogenesis, both FSH and testos-terone are necessary.
Negative Feedback Control of Seminiferous Tubule Activity— Role of the Hormone Inhibin.When the seminiferoustubules fail to produce sperm, secretion of FSH by the anterior pituitary gland increases markedly. Con-versely, when spermatogenesis proceeds too rapidly, pituitary secretion of FSH diminishes.The cause of this negative feedback effect on the anterior pituitary is believed to be secretion by the Sertoli cells of still another hormone called inhibin (see Figure 80–10).
This hormone has a strong direct effect on the ante-rior pituitary gland to inhibit the secretion of FSH and possibly a slight effect on the hypothalamus to inhibit secretion of GnRH.
Inhibin is a glycoprotein, like both LH and FSH, having a molecular weight between 10,000 and 30,000.It has been isolated from cultured Sertoli cells. Its potent inhibitory feedback effect on the anterior pitu- itary gland provides an important negative feedback mechanism for control of spermatogenesis, operating simultaneously with and in parallel to the negative feedback mechanism for control of testosterone secretion.
Psychic Factors That Affect Gonadotropin Secretion and Sexual Activity
Many psychic factors, feeding especially from the limbic system of the brain into the hypothalamus, can affect the rate of secretion of GnRH by the hypothal-amus and therefore can also affect most other aspects of sexual and reproductive functions in both the male and the female. For instance, transporting a prize bull in a rough truck is said to inhibit the bull’s fertility— and the human male is hardly different.
Human Chorionic Gonadotropin Secreted by the Placenta During Pregnancy Stimulates Testosterone Secretion by the Fetal Testes
During pregnancy, the hormone human chorionicgonadotropin (hCG) is secreted by the placenta, andit circulates both in the mother and in the fetus. This hormone has almost the same effects on the sexual organs as LH.
During pregnancy, if the fetus is a male, hCG from the placenta causes the testes of the fetus to secrete testosterone. This testosterone is critical for promoting formation of the male sexual organs, as pointed out earlier. We discuss hCG and its functions during pregnancy in greater detail.
Puberty and Regulation of Its Onset
Initiation of the onset of puberty has long been a mystery. But it has now been determined that duringchildhood the hypothalamus simply does not secrete significant amounts of GnRH. One of the reasonsfor this is that, during childhood, the slightest secretion of any sex steroid hormones exerts a strong inhibitory effect on hypothalamic secretion of GnRH. Yet, for reasons still not understood, at the time of puberty, the secretion of hypothalamic GnRH breaks through the childhood inhibition, and adult sexual life begins.
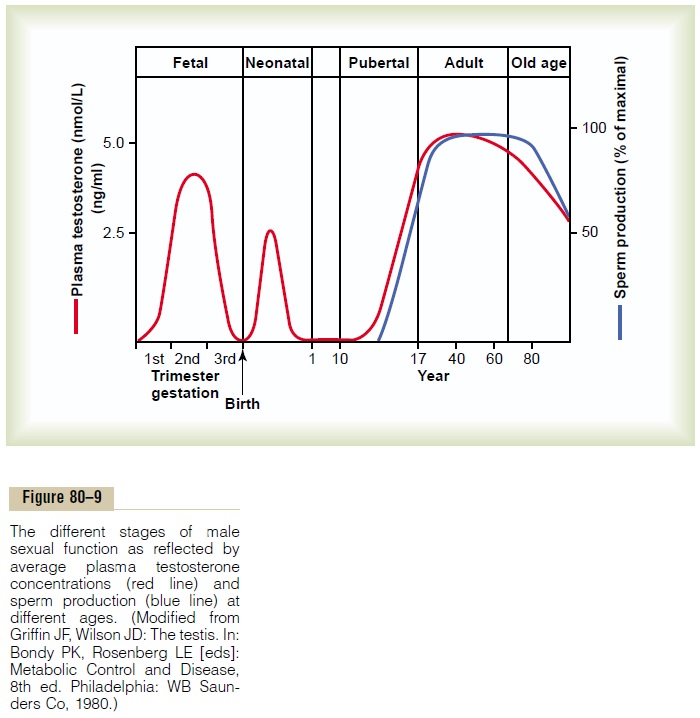
Male Adult Sexual Life and Male Climacteric. After puberty,gonadotropic hormones are produced by the male pitu-itary gland for the remainder of life, and at least some spermatogenesis usually continues until death. Most men, however, begin to exhibit slowly decreasing sexual functions in their late 40s or 50s, and one study showed that the average age for terminating intersexual rela-tions was 68, although the variation was great. This decline in sexual function is related to decrease in testosterone secretion, as shown in Figure 80–9. The decrease in male sexual function is called the male cli-macteric. Occasionally the male climacteric is associatedwith symptoms of hot flashes, suffocation, and psychic disorders similar to the menopausal symptoms of the female. These symptoms can be abrogated by adminis-tration of testosterone, synthetic androgens, or even estrogens that are used for treatment of menopausal symptoms in the female.
Related Topics