Chapter: Plant Biochemistry: Mitochondria are the power station of the cell
Compartmentation of mitochondrial metabolism requires specific membrane translocators
Compartmentation of mitochondrial metabolism requires specific membrane translocators
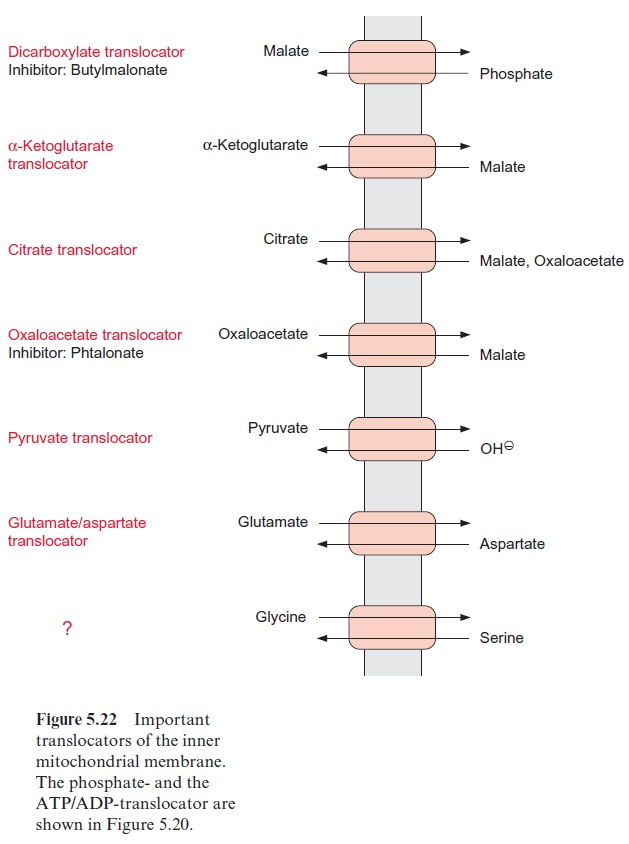
The mitochondrial inner membrane is impermeable for metabolites. Specific translocators enable a specific transport of metabolites between the mito-chondrial matrix and the cytosol in a counter-exchange mode (Fig. 5.22).
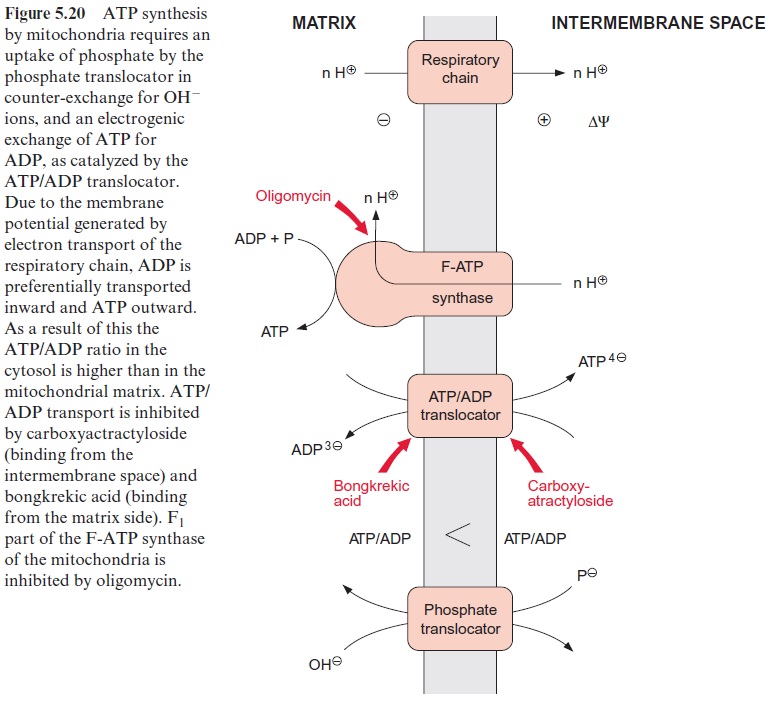
The role of the ATP/ADP and the phosphate translocators (Fig. 5.20) has been discussed. Malate and succinate are transported into the mitochondria in counter-exchange for phosphate by a dicarboxylate trans-locator. This transport is inhibited by butylmalonate. α-Ketoglutarate, cit-rate, and oxaloacetate are transported in counter-exchange for malate. By these translocators, substrates can be fed into the citrate cycle. Glutamate is transported in counter-exchange for aspartate, and pyruvate in counter-exchange for OH- ions. Although these translocators all occur in plant mitochondria, most of our present knowledge about them is based on stud-ies with mitochondria from animal tissues. A comparison of the amino acid sequences known for the ATP/ADP, phosphate, citrate, and glutamate/ aspartate translocators shows that they are homologous; the proteins of these translocators represent a family deriving from a common ancestor. All these translocators are composed of 2 x 6 transmembrane helices.
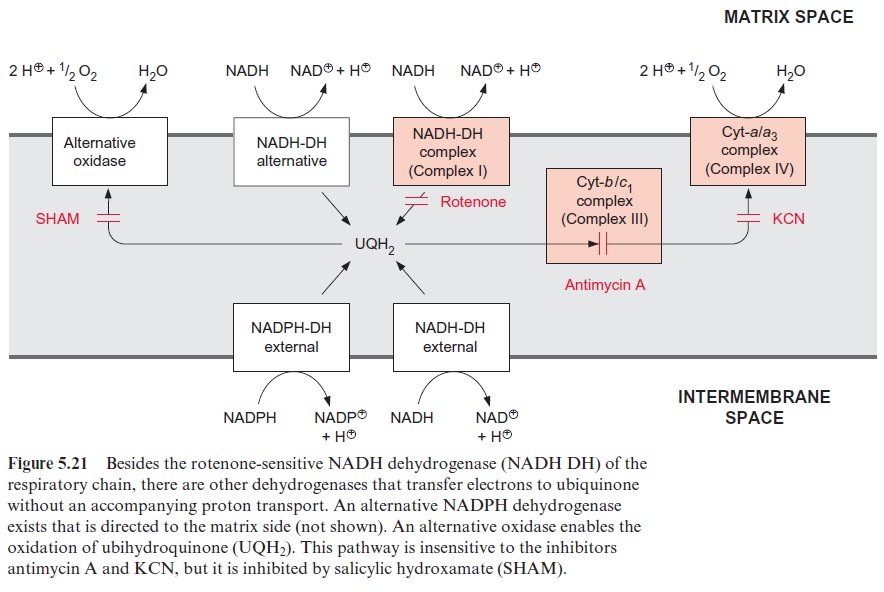
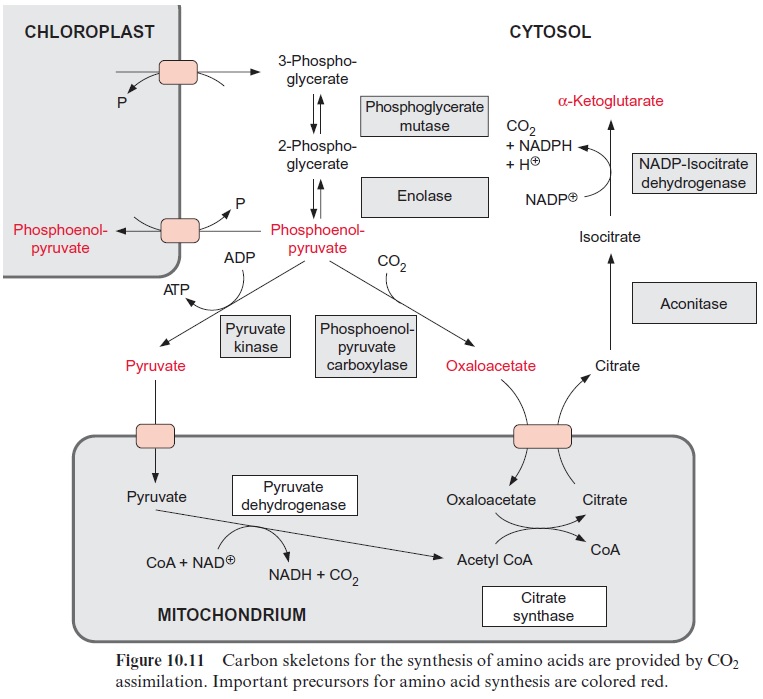
The malate-oxaloacetate translocator is a special component of plant mitochondria and has an important function in the malate-oxaloacetate cycle. It also transports citrate and is involved in providing the carbon skeletons for nitrate assimilation (Fig. 10.11). The oxaloacetate translocator and, to a lesser extent, the -ketoglutarate translocator are inhibited by the dicarboxylate phthalonate. The transport of glycine and serine, involved in the photorespiratory pathway , has not yet been characterized. Although final proof is still lacking, it is expected that this transport is mediated by one or two mitochondrial translocators.
Related Topics