Chapter: Medical Surgical Nursing: Management of Patients With Complications From Heart Disease
Cardiogenic Shock
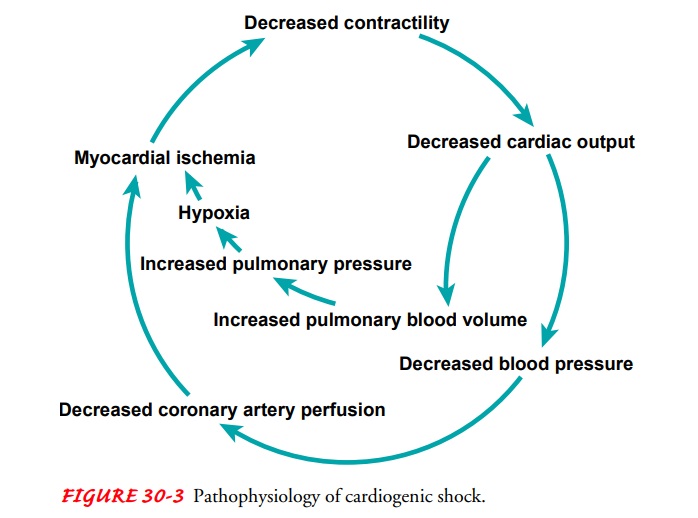
CARDIOGENIC SHOCK
Cardiogenic
shock occurs when the heart cannot pump enough blood to supply the amount of
oxygen needed by the tissues. This may occur because of one significant or
multiple smaller infarctions in which more than 40% of the myocardium be-comes
necrotic, because of a ruptured ventricle, significant valvular dysfunction,
trauma to the heart resulting in myocar-dial contusion, or as the end stage of
HF. It also can occur with cardiac tamponade, pulmonary embolism,
cardiomyopathy, and dysrhythmias.
Pathophysiology
The
signs and symptoms of cardiogenic shock reflect the circular nature of the
pathophysiology of HF. The degree of shock is pro-portional to the extent of
left ventricular dysfunction. The heart muscle loses its contractile power,
resulting in a marked reduc-tion in SV and CO, which is sometimes called
forward failure. The damage to the myocardium results in a decrease in CO,
which reduces arterial blood pressure and tissue perfusion in the vital organs
(heart, brain, lung, kidneys). Flow to the coronary arteries is reduced,
resulting in decreased oxygen supply to the myocardium, which increases
ischemia and further reduces the heart’s ability to pump. The inadequate
emptying of the ven-tricle also leads to increased pulmonary pressures,
pulmonary congestion, and pulmonary edema, exacerbating the hypoxia, causing
ischemia of vital organs, and setting a vicious cycle in motion (Fig. 30-3).
Clinical Manifestations
The
classic signs of cardiogenic shock are tissue hypoperfusion manifested as
cerebral hypoxia (restlessness, confusion, agitation), low blood pressure,
rapid and weak pulse, cold and clammy skin, increased respiratory crackles,
hypoactive bowel sounds, and decreased urinary output. Initially, arterial
blood gas analysis may show respiratory alkalosis. Dysrhythmias are common and
result from a decrease in oxygen to the myocardium.
Assessment and Diagnostic Findings
Use of
a PA catheter to measure left ventricular pressures and CO is important in
assessing the severity of the problem and planning management. The PA wedge
pressure is elevated and the CO decreased as the left ventricle loses its
ability to pump. The systemic vascular resistance is elevated because of the
sympathetic nervous system stimulation that occurs as a compensatory response
to the decrease in blood pressure. The decreased blood flow to the kidneys
causes a hormonal response (ie, increased catecholamines and activation of the
renin-angiotensin-aldosterone system) that causes fluid retention and further
vasoconstriction. Increases inHR, circulating volume, and vasoconstriction
occur to maintain circulation to the brain, heart, kidneys, and lungs, but at a
cost: an increase in the workload of the heart.
The
reduction in blood volume delivered to the tissues results in an increase in
the amount of oxygen that is extracted from the blood that is delivered to the
tissues (to try to meet the cellular demand for oxygen). The increased systemic
oxygen extraction results in decreased venous (mixed and central) oxygen
saturation. When the cellular oxygen needs cannot be met by the systemic oxygen
delivery and the oxygen extraction, anaerobic metabolism and the resulting
build up of lactic acid occur. Continuous central venous oximetry and
measurement of blood lactic acid levels may assist in assessing the severity of
the shock as well as the effectiveness of
treatment
Continued
cellular hypoperfusion eventually results in organ failure. The patient becomes
unresponsive, severe hypotension ensues, and the patient develops shallow
respirations; cold, cyanotic or mottled skin; and absent bowel sounds. Arterial
blood gas analysis shows metabolic acidosis, and all laboratory test results
indicate organ dysfunction.
Medical Management
The
major approach to treating cardiogenic shock is to correct the underlying
problems, reduce any further demand on the heart, im-prove oxygenation, and
restore tissue perfusion. For example, if the ventricular failure is the result
of an acute myocardial infarction, emergency percutaneous coronary intervention
may be indicated (Webb et al., 2001). Ventricular assist devices may be
implanted to support the pumping action of the heart (Barron et al., 2001).
Major dysrhythmias are corrected because they may have caused or contributed to
the shock. If the patient has hypervolemia, diuresis is indicated. Diuretics,
vasodilators, and me-chanical devices, such as filtration (continuous renal
replacement therapy [CRRT]) and dialysis, have been used to reduce the
circu-lating blood volume. If hypovolemia or low intravascular volume is
suspected or detected through pressure readings, the patient is given
intravenous volume expanders (eg, normal saline solution, lactated Ringer’s
solution, albumin) to increase the amount of cir-culating fluid. The patient is
placed on strict bedrest to conserve energy. If the patient has hypoxemia, as
detected by pulse oxime-try or arterial blood gas analysis, oxygen
administration is increased, often under positive pressure when regular flow is
insufficient to meet tissue demands. Intubation and sedation may be necessary
to maintain oxygenation. The settings for mechanical ventilation are adjusted
according to the patient’s oxygenation status and the need for conserving
energy.
PHARMACOLOGIC THERAPY
Medication
therapy is selected and guided according to CO, other cardiac parameters, and
mean arterial blood pressure. Because of the decreased perfusion to the
gastrointestinal system and the need to adjust the dosage quickly, most
medications are administered intravenously.
Vasopressors,
or pressor agents, are medications used to raise blood pressure and increase
CO. Many pressor medications are catecholamines, such as norepinephrine
(Levophed) and high-dose (>10 μg/kg per minute)
dopamine (Intropin). Their pur-pose is to promote perfusion to the heart and
brain, but they compromise circulation to other organs (eg, kidney). Because
they also tend to increase the workload of the heart by increasing oxygen
demand, they are not administered early in the cardio-genic shock process.
Diuretics
and vasodilators may be administered carefully to reduce the workload of the
heart as long as they do not cause worsening of the tissue hypoperfusion.
Agents such as amrinone (Inocor), milrinone (Primacor), sodium nitroprusside
(Nipride), and nitroglycerin (Tridil) are effective vasoactive medications that
lower the volume returning to the heart, decrease blood pressure, and decrease
cardiac work. They cause the arteries and veins to dilate, thereby shunting
much of the intravascular volume to the periphery and causing a reduction in
preload and afterload.
Positive
inotropic medications are given to increase myocardial contractility. Dopamine
(Intropin, given at more than 2 μ g/kg per minute),
dobutamine (Dobutrex), and epinephrine (Adrenalin) are catecholamines that
increase contractility. Each of these can cause tachydysrhythmias because they
increase automaticity with increasing dosage. Monitoring baseline HR is
therefore impor-tant. As the baseline HR increases, so does the risk of
developing tachydysrhythmias.
OTHER TREATMENTS
Other
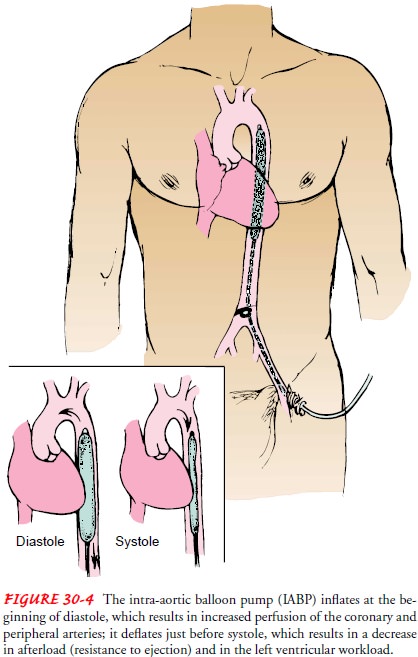
therapeutic modalities for cardiogenic shock include use of circulatory assist
devices. The most frequently used mechanical support device is the intra-aortic
balloon pump (IABP). The IABP is a catheter with an inflatable balloon at the
end. The catheter is usually inserted through the femoral artery, and the
balloon is po-sitioned in the descending thoracic aorta (Fig. 30-4). IABP uses
internal counterpulsation through the regular inflation and defla-tion of the
balloon to augment the pumping action of the heart. The device inflates during
diastole, increasing the pressure in the aorta during diastole and therefore
increasing blood flow through the coronary and peripheral arteries. It deflates
just before systole, lessening the pressure within the aorta before left
ventricular con-traction, decreasing the amount of resistance the heart has to
over-come to eject blood and therefore decreasing the amount of work the heart
must put forth to eject blood. The device is connected to a console that
synchronizes the inflation and deflation of the balloon with the ECG or the
arterial pressure (as indicators for sys-tole and diastole). Hemodynamic
monitoring is essential to de-termine the patient’s response to the IABP.
Nursing Management
The
patient in cardiogenic shock requires constant monitoring and intensive care.
The critical care (intensive care) nurse must carefully assess the patient,
observe the cardiac rhythm, monitor hemodynamic parameters, and record fluid
intake and urinary output. The patient must be closely assessed for responses
to the medical interventions and for the development of complications, which
must be corrected immediately.
Because
of the frequency of nursing interventions and the technology required for
effective medical management, the pa-tient is always treated in an intensive
care environment. Critical care nurses are responsible for the nursing
management, which includes frequent assessments and timely adjustments to
medica-tions and therapies based on the assessment data.
Related Topics