Electricity | Chapter 5 | 8th Science - Atom | 8th Science : Chapter 5 : Electricity
Chapter: 8th Science : Chapter 5 : Electricity
Atom
Atom
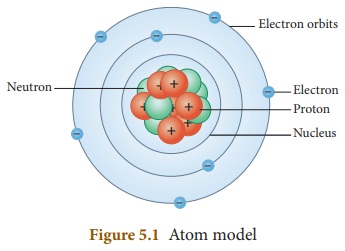
An atom consists of proton, electron
and neutron which are called sub-atomic particles. Proton and neutron are found
inside the nucleus which is at the centre of an atom. Electrons revolve around
the nucleus in different paths called orbits. In an atom, the number of protons
and the number of electrons will be equal. There is a force of attraction
between the protons in the nucleus and the electrons in the orbits. Electrons
in the inner orbits are strongly attracted by the protons and they cannot be
removed from the atom easily. But, the electrons in the outermost orbits are
loosely bound and they can be easily removed from the atom.
Related Topics