Chapter: Modern Medical Toxicology: Chemical Poisons: Heavy Metals
Arsenic - Chemical Poisons

Arsenic
Arsenic
is thought to occur throughout the universe. It is the twentieth most common
element in the earth’s crust, having a concentration of 1.8 ppm. Arsenic is
today the commonest source of acute heavy metal poisoning, and is second only
to lead in the incidence of chronic toxicity.
Physical Appearance
·
Arsenic is a metalloid i.e. it is an
element which resembles a metal in some respects, and is by itself not very
toxic.
·
However, almost all the salts are
toxic to varying degree. Arsenic is a silver-grey or tin-white, shiny, brittle,
crystal- line and metallic-looking element (Fig 9.1). It is rarely found in its isolated, elemental form. More
commonly, it is present in mineral species, in alloys, or as an oxide or other
compound form.

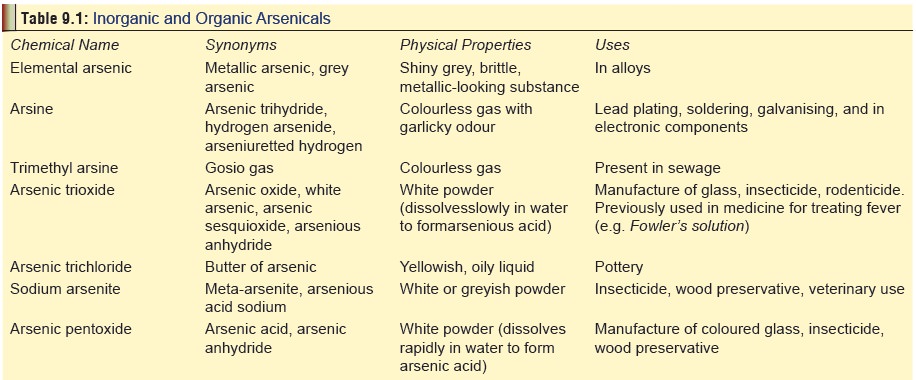
· Table 9.1 displays the physical properties and uses of arsenic and its compounds.

Usual Fatal Dose
·
200 to 300 mg for arsenic trioxide.
·
In general, the pentavalent form of
arsenic (arsenate) is less toxic than
the trivalent form (arsenite) because
it is less water soluble.
·
The most toxic form is arsine gas
(25 to 30 ppm can be lethal in 30 minutes).
Toxicokinetics and Mode of Action
·
Arsenic is absorbed through all
portals of entry including oral, inhalational, and cutaneous routes.
·
After absorption it is redistributed
to the liver, lungs, intestinal wall, and spleen, where it binds to the
sulfydryl groups of tissue proteins. Arsenic replaces phosphorus in the bone
where it may remain for years. It gets deposited also in hair.
·
While arsenic does not cross the
blood-brain barrier easily, it crosses the placenta readily and can give rise
to intrauterine death of the foetus. In less severe intoxications it can cause
respiratory distress of the newborn due to pulmonary haemorrhage and hyaline
membrane formation.
Clinical Features
·
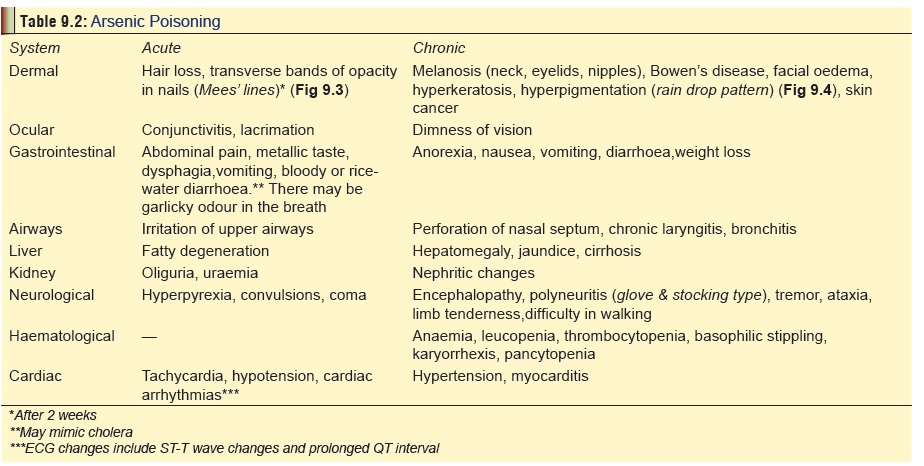
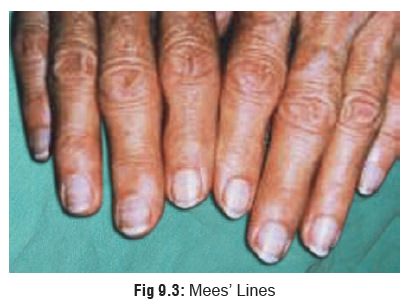
Mentioned in Table 9.2.
·
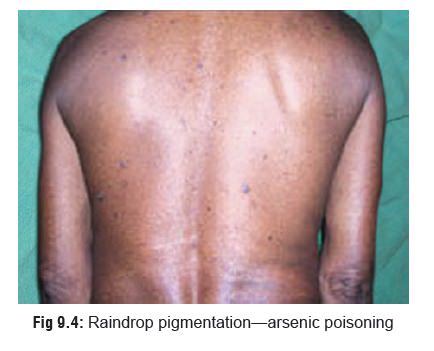
Dermal pigmentation is consistently
seen in chronic arsenic poisoning, and may also be encountered in exposure to
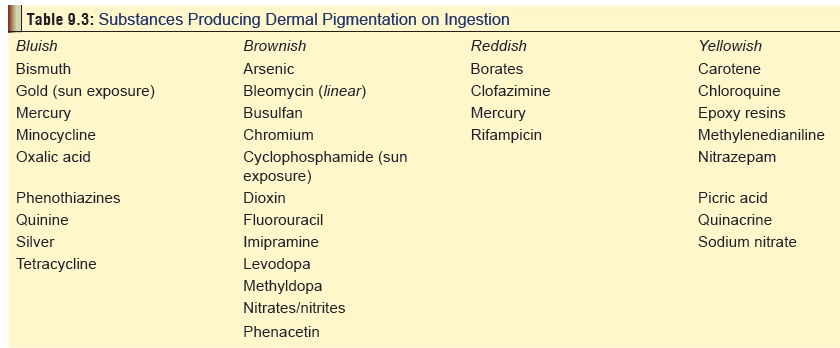
certain other substances, (Table 9.3).
·
In West Bengal, thousands of people
residing in more than villages are known to consume
arsenic-contaminated groundwater, and many of them have arsenical skin lesions.
Conjunctivitis, depigmented lesions, melanosis, and hyper-keratosis (Fig 9.2), are most commony present, but
malignant neoplasms and gangrenous lesions have also been found. Pulmonary
lesions are also fairly common.

Diagnosis
·
Urine level: If the 24 hour excretion of arsenic
exceeds100 mcg, it is indicative of toxicity. However, ingestion of seafood can
interfere with interpretation since consid-erable concentrations of organic
arsenicals such as arse-nobetaine and arsenocholine may be present in
shellfish,cod, haddock, etc., although it is not associated with toxic
·
Blood level: This is less reliable than urine
level becauseof short half-life of arsenic in the blood. However, a blood level
of arsenic less than 7 mcg/100 mL (70 mcg/L) is generally considered in the
normal range.
· Hair level: Although considered to be an important diag-nostic criterion, it is actually virtually useless since it cannot discriminate between external deposition and toxic accumulation. If hair is sent for arsenic quantitation, pubic hair instead of scalp hair should be sent because of the possibility of scalp hair being contaminated with arsenic from the environment.
·
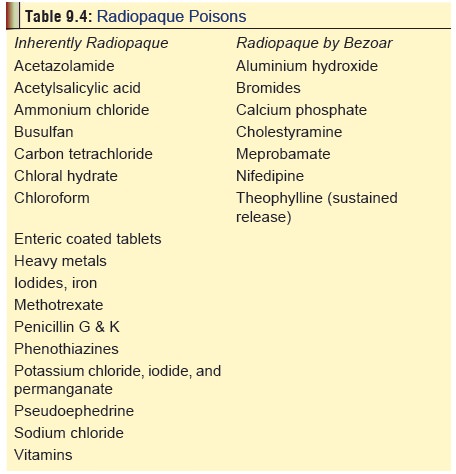
Radiography: Since arsenic is radiopaque,
abdominal x-ray may reveal its presence in the gastrointestinal tract in acute
poisoning. Table 9.4 presents a list
of radiopaque poisons commonly encountered in practice.
·
Additional
investigations:
Monitor CBC, serum electrolytes, urinalysis (for
proteinuria, haematuria or pyuria), liver and renal func- tion tests. Obtain an
ECG and institute continuous cardiac monitoring in symptomatic patients.
Obtain a chest radiograph in patients with severe poisoning
or pulmonary effects.
Initial and periodic biological monitoring and medical
surveillance are required for employees exposed to arsenic.
Treatment
·
Supportive
measures: gastric lavage, intravenous fluids, cardiac monitoring,
etc.
·
Chelation
therapy: This can be done with BAL (BritishAnti Lewisite or
dimercaprol), penicillamine, DMSA (Dimercapto succinic acid), or DMPS
(Dimercapto propane sulfonic acid).
·
The usual agent employed is BAL at a
dose of 3 to 5 mg/kg intramuscularly every 4 hours until the urinary arsenic
excretion dips below 50 mcg/24 hours. Usual duration of therapy is 7 to 10
days.
·
In patients who are not allergic to
penicillin, penicil-lamine can be given orally at a dose of 100 mg/kg/day, 6th
hourly for 5 days.
·
DMSA and DMPS said to be superior to
BAL and penicillamine, are currently not available in India.
Principles of chelation:
·
Begin chelation therapy in
symptomatic patients. The urine arsenic level which should prompt chela-tion in
an asymptomatic patient has been recom-mended as 200 mcg/litre.
·
Repeat courses of chelation therapy
should be prescribed in severe poisonings until the 24-hour urine arsenic level
falls below 50 mcg/litre. Observation for return of symptoms is strongly
recommended.
·
Chelation therapy is not very
effective for chronic poisoning, and is totally ineffective in arsine
poisoning. The latter should be treated with emphasis on respiratory
stabilisation and haemo-dialysis.
Haemodialysis
or exchange transfusion.
Autopsy Features
·
Gastrointestinal congestion is a noteworthy feature in acute
arsenic poisoning and varies from a mere reddening of mucosa (red velvet) to frank haemorrhagic
gastritis. Focal haemorrhages giving rise to a flea bitten appearance is said to be characteristic. The intestines
may be inflamed and may contain “rice water” contents.![]()
·
Subendocardial haemorrhages are
often seen in the heart.
·
There may be evidence of fatty
degeneration of heart, liver, and kidneys.
·
Chronic arsenic poisoning may
present features of non-specific gastrointestinal inflammation, as well as
renal and hepatic damage, at autopsy.
·
It is conventional to preserve apart
from the routine viscera and body fluids, a piece of long bone (preferably femur), a bunch of pulled scalp hair, a wedge of
muscle, and a small portion of skin (from the back of the trunk) for chemical
analysis.
Forensic Issues
■■ Arsenic
has had an outstanding reputation as an ideal homicidal poison especially in
the West, particularly United Kingdom in the Victorian era. Several celebrated
murders are said to have been accomplished with the help of arsenic during this
period. One of the most shocking cases which remained speculative until
recently, when scientific evidence finally established the truth beyond
reasonable doubt, was that of Napoleon Bonaparte.
■■ Arsenic
trioxide being almost tasteless and colourless in solution can be administered
without arousing the suspicion of a victim. The main obstacle is relative
insolubility. But the solubility can be greatly enhanced if hot solutions are
used such as coffee, tea, cocoa, porridge or gruel, and soups. The only problem
is that on cooling, much of the dissolved arsenic will separate out to yield a
gritty deposit. On chronic successful administration to a victim, arsenic
produces insidious but relentlessly progressive symptoms which are likely to be
mistaken even by a medical practitioner for natural causes such as neurological
disease, alcoholism, tuberculosis, and hepatic or renal afflictions, while
acute poisoning may be confused with gastroenteritis or cholera.
■■ However
the popularity of arsenic has declined in recent times because of various
factors, and today most cases of arsenic poisoning are accidental, though
murders are still reported from time to time. Accidental poisoning may be the
result of industrial or occupational exposure, or it may be due to consumption
of contaminated water or food.
·
In the past, certain allopathic drugs used to contain
arsenic and iatrogenic poisoning was not uncommon. Today ayurvedic preparations
constitute the main hazard, some of which can contain substantial
concen-trations of arsenic that can produce chronic poisoning on prolonged use.
· One of the most important sources of chronic acci-dental poisoning in India is consumption of well water. There are indications that sizeable populations of several Asian countries are exposed to arsenic tainted water, particularly tube well water. A devastating health crisis (endemic hydroarsenicism) began to unfold in West Bengal in the early 1980s due to exposure to arsenic laced well water.
High levels of arsenic have been demonstrated which probably
leached from natural underground sources into thousands of village wells
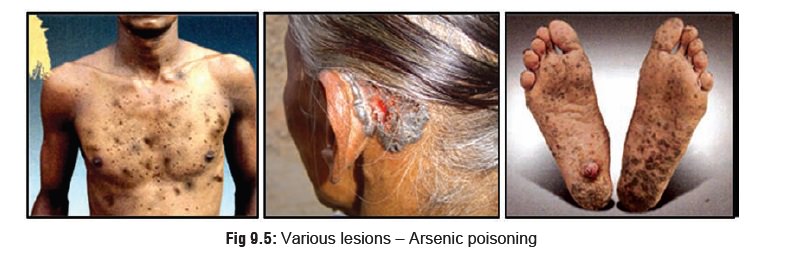
affecting more than 250,000 people who display overt manifestations (especially
related to the skin) such as hyperkeratosis, pigmentation, and skin cancer (Fig 9.5), while more than 1 million who
are continuing to drink the tainted water may develop lesions over a period of
time. While the exact cause for this wide-spread contamination of water could
not be pinpointed, it is believed that the problem is related to the large
scale withdrawal of ground water. Whatever the under-lying mechanism, the fact
remains that analysis of water from more than 200,000 tube wells demonstrated
arsenic content above the WHO permissible limit of 0.01 mg/L in more than 60%
samples (some as high as 3.7 mg/L). Hair, urine, skin, and nail samples from
people drinking the water have also been shown to contain high levels of arsenic.
In order to counter this tragedy, the West Bengal government is in the process
of arranging the supply of arsenic-free piped water from the Ganges river, but
the full implementation of such a programme may take several years, and involve
the expenditure of millions of dollars of foreign aid. The most alarming
question raised by this tragedy is how many tens of millions of people may be
exposed to high levels of arsenic in areas not yet tested for contamination
elsewhere in India.
Related Topics