Chapter: Medical Immunology: Tolerance and Autoimmunity
Animal Models of Autoimmunity
Animal Models of Autoimmunity
Our understanding of autoimmune disease has been facilitated by studies in animal models. Several animal models have been developed, each sharing some characteristics of a human disease of autoimmune etiology. These animal models often provide the only experi-mental approaches to the study of the pathogenesis of autoimmune diseases.
In some experimental models, injecting normal animals with antigens extracted from the human target tissues induces autoimmune diseases. A rapid onset and an acute course characterize the resulting diseases. These models have been particularly useful in the study of autoimmune thyroiditis and arthritis (collagen-induced arthritis). Most useful for the study of autoimmunity are animals that develop autoimmune disease spontaneously, whose course is protracted and parallels closely the disease as seen in humans. Representative an-imal models of different autoimmune diseases are listed in Table 16.10.
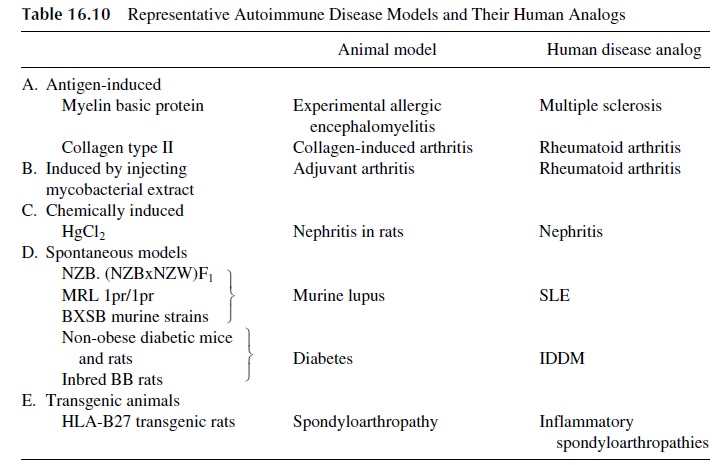
1. Experimental Allergic Encephalomyelitis
Experimental allergic encephalomyelitis (EAE) in mice and rats is the best characterized experimental model of organ-specific autoimmune disease. Immunizing animals with myelin basic protein and adjuvant induces the disease. One to 2 weeks later, the animals develop encephalomyelitis characterized by perivascular mononuclear cell infiltrates and demyelination. The mononuclear cell infiltrates show a predominance of CD4+ helper T lymphocytes, which upon activation release cytokines that attract phagocytic cells to the area of immunological reaction; those cells are, in turn, activated and release enzymes re-sponsible for the demyelination. CD4+ T-lymphocyte clones from animals with EAE dis-ease can transfer the disease to normal animals of the same strain. Genetic manipulations leading to deletion of the genes coding for two specific variable regions of the TcR β chain (Vβ8 and Vβ13) prevent the expression of disease. These two Vβ regions must obviously be involved in the recognition of a dominant epitope of human myelin.
2. Diabetes
Diabetes develops spontaneously in inbred BB rats, as well as in nonobese diabetic (NOD) mice. In both strains the onset of the disease is characterized by T-cell–mediated insulitis, which evolves into diabetes. This disease demonstrates H-2 linkage remarkably similar to that observed between human IDDM and HLA-DR3, DR4, and other MHC-II alleles. In NOD mice, a decreased expression of MHC-I genes, secondary to a TAP-1 gene defi-ciency, has been recently characterized. Such a deficiency would prevent these animals from deleting autoreactive clones during T-cell differentiation.
3. Systemic Lupus Erythematosus
A number of murine strains spontaneously develop autoimmune disease that resembles hu-man SLE:
· (NZB×NZW)F1 female mice develop glomerulonephritis, hemolytic anemia, and anti-DNA antibodies. Numerous alterations in T- and B-lymphocyte function, cytokine release, and macrophage functions have been described in these ani-mals.
· MRL-Ipr/Ipr mice, which lack Fas antigen, and gld mice, which lack Fas ligand, pro-duce autoantibodies and develop arthritis and kidney disease, but they also de-velop massive lymphadenopathy, which is not seen in human disease.
· BXSB mice develop anti-DNA antibodies, nephritis, and vasculitis. In this strain, in contrast to the others, disease susceptibility is linked to the Y chromosome.
Related Topics