Chapter: Organic Chemistry: Alkyl halides
Alkyl halides: Elimination
ELIMINATION
Key Notes
Definition
Alkyl
halides undergo elimination reactions with nucleophiles or bases, where
hydrogen halide is lost from the molecule to produce an alkene.
There
are two commonly occurring mechanisms. The E2 mechanism is the most effective
for the synthesis of alkenes from alkyl halides and can be used on primary,
secondary, and tertiary alkyl halides. The E1 reaction is not particularly
useful from a synthetic point of view and occurs in compe- tition with the SN1
reaction. Tertiary alkyl halides and some secondary alkyl halides can react by
this mechanism, but not primary alkyl halides.
Susceptible β-protons
Elimination
is possible if the alkyl halide contains a susceptible β-proton which can be
lost during the elimination reaction. β-Protons are situated on the carbon
linked to the carbon of the C–X group.
E2 Mechanism
The E2
mechanism is a concerted, one-stage process involving both alkyl halide and
nucleophile. The reaction is second order and depends on the concentration of
both reactants.
E1 Mechanism
The E1
mechanism is a two-stage process involving loss of the halide ion to form a
carbocation, followed by loss of the susceptible proton to form the alkene. The
rate determining step is the first stage involving loss of the halide ion. As a
result, the reaction is first order, depending on the concen- tration of the
alkyl halide alone. The carbocation intermediate is stabilized by substituent
alkyl groups.
E2 versus E1
The E2
reaction is more useful than the E1 reaction in synthesizing alkenes. The use
of a strong base in a protic solvent favors the E2 elimination over the E1
elimination. The E1 reaction occurs when tertiary alkyl halides are dissolved
in protic solvents.
Definition
Alkyl halides which have a proton attached to a
neighboring β-carbon atomcan undergo an elimination reaction
to produce an alkene plus a hydrogenhalide
(Fig. 1). In
essence, this reaction
is the reverse
of the electrophilicaddition of a hydrogen halide to
an alkene.
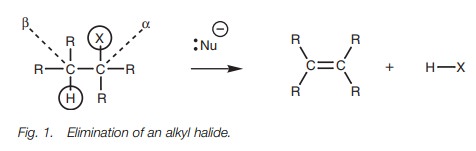
There are twomechanisms by which this
elimination can take place – the E2 mechanism andthe E1 mechanism.
The E2 reaction is the most effective for the synthesis of alkenes from alkyl halides and can be used on primary, secondary, and tertiary alkyl halides. The E1reaction is not particularly useful from a synthetic point of view and occurs in competition with the SN1 reaction of tertiary alkyl halides. Primary and secondary alkyl halides do not usually react by this mechanism.
Susceptible
β-protons
An alkyl halide can undergo an elimination
reaction if it has a susceptible proton situated on a β-carbon, that is, the carbon next to the C–X group. This proton is
lost during the elimination reaction along with the halide ion. In some
respects, there is a similarity here between alkyl halides and carbonyl
compounds (Fig. 2). Alkyl halides can
have susceptible protons at the β-position whilst carbonyl com-pounds can have
acidic protons at their α-position. If we compare both structures, we
can see that the acidic/susceptible proton is attached to a carbon neighboring
an electrophilic carbon.

E2 Mechanism
The E2 mechanism is a concerted mechanism
involving both the alkyl halide and the nucleophile. As a result, the reaction
rate depends on the concentration of both reagents and is defined as second
order (E2 = Elimination second order). To illustrate the mechanism, we shall
look at the reaction of 2-bromopropane with a hydroxide ion (Fig. 3).
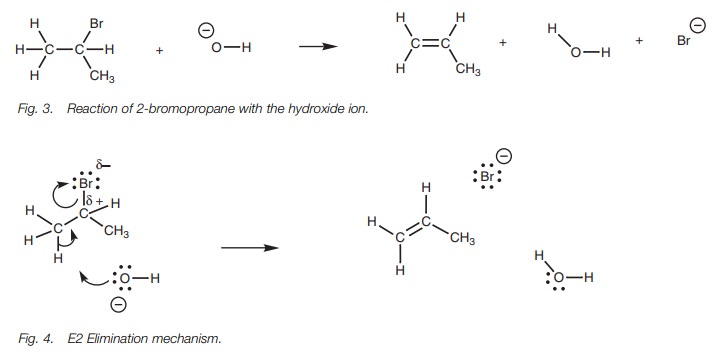
The mechanism (Fig. 4) involves the hydroxide ion forming a bond to the
susceptible proton. As the hydroxide ion forms its bond, the C–H bond breaks.
Both electrons in that bond could move onto the carbon, but there is a
neighbor-ing electrophilic carbon which attracts the electrons and so the
electrons move in to form a π bond between the two carbons. At the same time
as this π bond is formed, the C–Br bond breaks and both
electrons end up on the bromine atom which is lost as a bromide ion.
The E2 elimination is stereospecific, with elimination occurring in an anti-periplanar geometry. The diagrams (Fig. 5) show that the four atoms involved in the reaction are in a plane with the H and Br on opposite sides of the molecule.
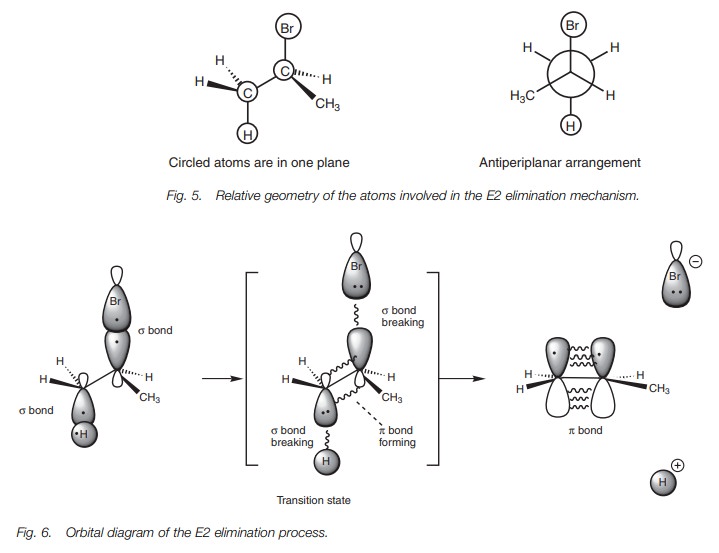
The reason for this stereospecificity can be
explained using orbital diagrams (Fig. 6).
In the transition state of this reaction, the C–H and C–Br σ bonds are in the process of breaking. As they do so, the sp3 hybridized orbitals which
were used for these σ bonds are changing into p orbitals which start to interact with each other to form the
eventual π bond. For all this to happen in the one
transition state, an antiperiplanar arrangement is essential.
E1 Mechanism
The E1 mechanism usually occurs when an alkyl halide is dissolved in a protic solvent where the solvent can act as a nonbasic nucleophile. These are the same conditions for the SN1 reaction and so both these reactions usually occur at the same time resulting in a mixture of products. As an example of the E1 mechanism, we shall look at the reaction of 2-iodo-2-methylbutane with methanol (Fig. 7).
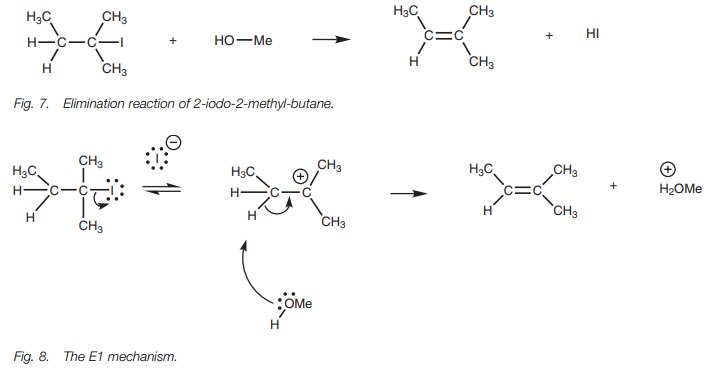
There are two stages to this mechanism (Fig. 8). The first stage is exactly the
same process described for the SN1 mechanism and that is cleavage of
the C–X bond to form a planar carbocation intermediate where the positive charge
is sta- bilized by the three alkyl groups surrounding it. In the second stage,
the methanol forms a bond to the susceptible proton on the β-carbon. The C–H bond breaks and both electrons are used to form a π bond to the neighboring carbocation. The first step of the
reaction mechanism is the rate-determining step and since this is only
dependent on the concentration of the alkyl halide, the reaction is first order
(El elimination first order). There is no stereospecificity involved in this
reaction and a mixture of isomers may be obtained with the more stable (more
substituted) alkene being favored.
E2 versus E1
The E2 elimination occurs with a strong base
(such as a hydroxide or ethoxide ion) in a protic solvent (such as ethanol or
water). The E2 reaction is more common than the E1 elimination and more useful.
All types of alkyl halide can undergo the E2 elimination and the method is
useful for preparing alkenes.
The conditions which suit E1 are the same which
suit the SN1 reaction (i.e. a pro- tic solvent and a nonbasic
nucleophile). Therefore, the E1 reaction normally only occurs with tertiary
alkyl halides and will be in competition with the SN1 reaction.
Related Topics