Chapter: Clinical Anesthesiology: Clinical Pharmacology: Adrenergic Agonists & Antagonists
Adrenoceptor Physiology
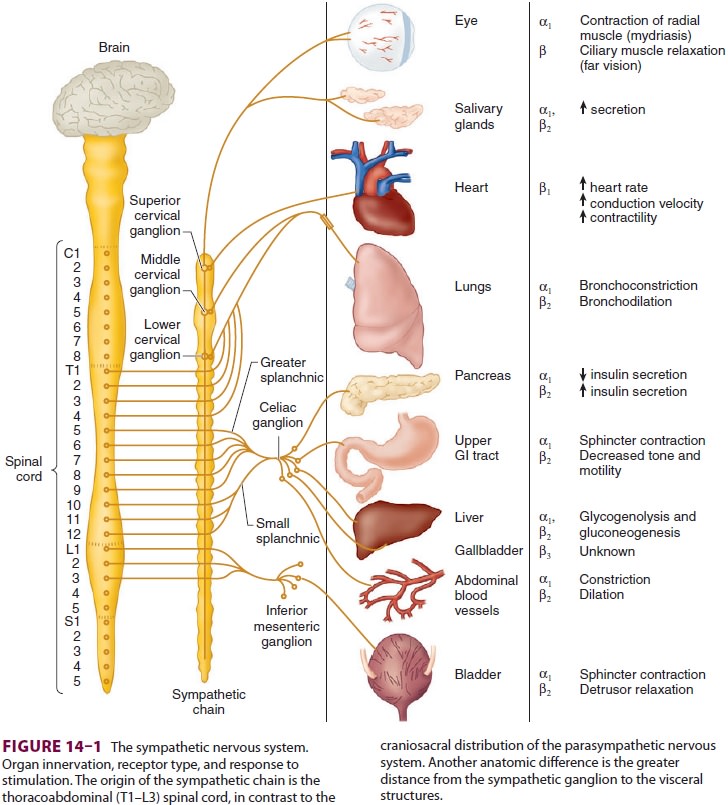
ADRENOCEPTOR PHYSIOLOGY
The
term “adrenergic” originally referred to the effects of epinephrine (adrenaline), although nor-epinephrine
(noradrenaline) is the primary neu-rotransmitter responsible for most of the
adrenergic activity of the sympathetic nervous system. With the exception of
eccrine sweat glands and some blood vessels, norepinephrine is released by
post-ganglionic sympathetic fibers at end-organ tissues (Figure 14–1). In contrast,
acetylcholine is released by preganglionic sympathetic fibers and all
para-sympathetic fibers.
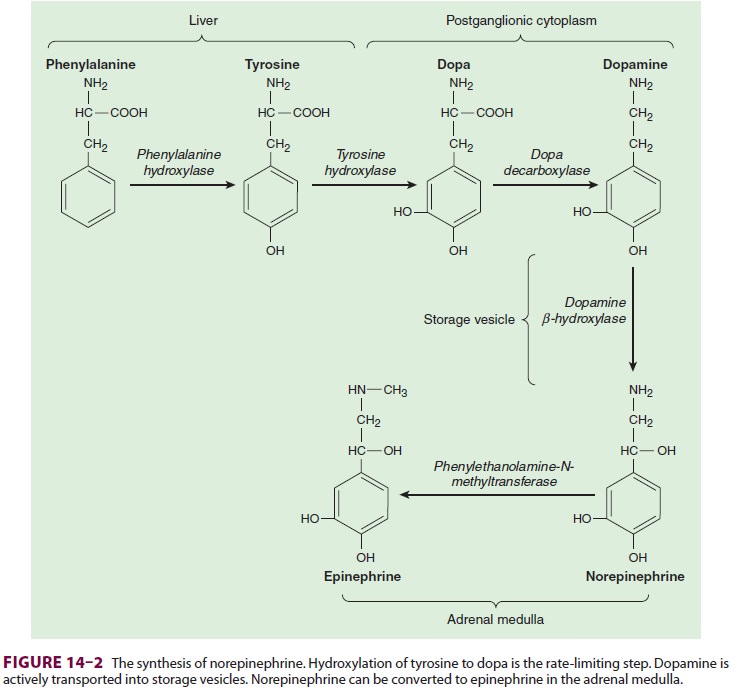
Norepinephrine
is synthesized in the cyto-plasm of sympathetic postganglionic nerve end-ings
and stored in the vesicles (Figure 14–2). After release by a process of
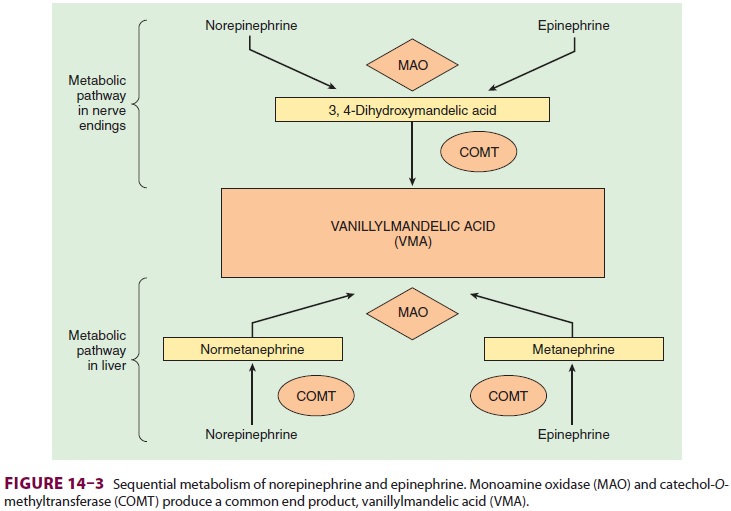
exocytosis, the action of norepinephrine is primarily terminated by reup-take
into the postganglionic nerve ending (inhib-ited by tricyclic antidepressants),
but also by diffusion from receptor sites, or via metabolism by monoamine
oxidase (inhibited by monoamine oxidase inhibitors) and catechol-O-methyltransfer-ase (Figure 14–3).
Prolonged adrenergic activation leads to desensitization and hyporesponsiveness
to further stimulation.
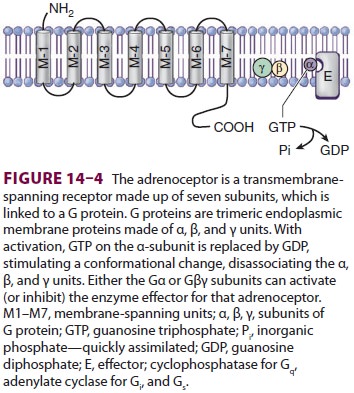
Adrenergic
receptors are divided into two gen-eral categories: α and β. Each of these has
been fur-ther subdivided into at least two subtypes: α1
and α2,
and β1, β2,
and β3.
The α-receptors
have beenfurther divided using molecular cloning techniques into α1A,
α1B,
α1D,
α2A,
α2B,
and α2C.
These receptors are linked to G proteins ( Figure 14–4; Drs. Rodbell and Gilman received
the Nobel Prize in physiology or medicine in 1994 for their
discovery)—heterotri-meric receptors with α, β, and γ subunits. The
differ-ent adrenoceptors are linked to specific G proteins, each with a unique
effector, but each using guano-sine triphosphate (GTP) as a cofactor. α1
is linked to Gq, which activates phospholipases; α2
is linked to Gi, which inhibits adenylate cyclase, and β
is linked to Gs, which activates adenylate cyclase.
α1-Receptors
α1-Receptors
are postsynaptic adrenoceptorslocated in smooth muscle throughout the body (in
the eye, lung, blood vessels, uterus, gut, and geni-tourinary system).
Activation of these receptors increases intracellular calcium ion
concentration, which leads to contraction of smooth muscles. Thus, α1-agonists
are associated with mydriasis (pupil-lary dilation due to contraction of the
radial eye muscles), bronchoconstriction, vasoconstriction, uterine
contraction, and constriction of sphincters in the gastrointestinal and
genitourinary tracts. α1-stimulation
also inhibits insulin secretion andlipolysis. The myocardium possesses α1-receptors
that have a positive inotropic effect, which might play a role in
catecholamine-induced arrhythmia. During myocardial ischemia, enhanced α1-receptor
coupling with agonists is observed. Nonetheless, the most important cardiovascular
effect of α1-stimulation
is vasoconstriction, which increases peripheral vascular resistance, left
ventricular after-load, and arterial blood pressure.
α2-Receptors
In
contrast to α1-receptors,
α2-receptors
are located primarily on the presynaptic nerve termi-nals. Activation of these
adrenoceptors inhibits adenylate cyclase activity. This decreases the entry of
calcium ions into the neuronal terminal, which limits subsequent exocytosis of
storage vesicles containing norepinephrine. Thus, α2-receptors
cre-ate a negative feedback loop that inhibits further norepinephrine release
from the neuron. In addi-tion, vascular smooth muscle contains postsyn-aptic α2-receptors
that produce vasoconstriction. More importantly, stimulation of postsynaptic α2-receptors
in the central nervous system causessedation and reduces sympathetic outflow,
which leads to peripheral vasodilation and lower blood pressure.
β1-Receptors
β-Adrenergic receptors are classified into β1, β2, and β3receptors. The catecholamines, norepinephrine, and epinephrine are equipotent on β1 receptors, but epinephrine is significantly more potent than nor-epinephrine on β2 receptors.The most important β1-receptors are located on the postsynaptic membranes in the heart. Stimulation of these receptors activates adenyl-ate cyclase, which converts adenosine triphosphate to cyclic adenosine monophosphate and initiates a kinase phosphorylation cascade. Initiation of the cascade has positive chronotropic (increased heart rate), dromotropic (increased conduction), and ino-tropic (increased contractility) effects.
β2-Receptors
β2-Receptors
are primarily postsynaptic adreno-ceptors located in smooth muscle and gland
cells. They share a common mechanism of action with β1-receptors:
adenylate cyclase activation. Despitethis commonality, β2
stimulation relaxes smooth muscle, resulting in bronchodilation, vasodilation,
and relaxation of the uterus (tocolysis), bladder, and gut. Glycogenolysis,
lipolysis, gluconeogenesis, and insulin release are stimulated by β2-receptor
activa-tion. β2-agonists
also activate the sodium–potassium pump, which drives potassium intracellularly
and can induce hypokalemia and dysrhythmias.
β3-Receptors
β3-Receptors are found in the gallbladder and brainadipose tissue. Their role in gallbladder physiology is unknown, but they are thought to play a role in lipolysis and thermogenesis in brown fat.
Dopaminergic Receptors
Dopamine (DA) receptors are a group of
adrener-gic receptors that are activated by dopamine; these receptors are
classified as D1 and D2
receptors. Acti-vation of D1 receptors mediates vasodilation in the
kidney, intestine, and heart. D2 receptors are believed to play a role
in the antiemetic action of droperidol.
Related Topics