Chapter: Medical Surgical Nursing: Management of Patients With Structural, Infectious, and Inflammatory Cardiac Disorders
Valve Replacement
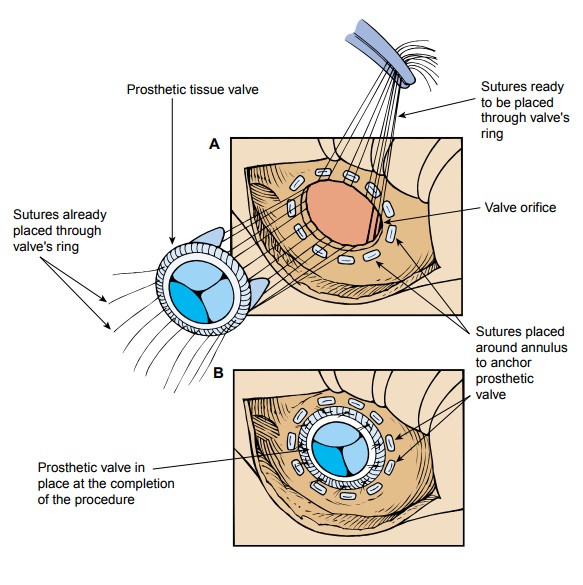
VALVE REPLACEMENT
Prosthetic
valve replacement began in the
1960s. When valvu-loplasty or valve repair is not a viable alternative, such as
when the annulus or leaflets of the valve are immobilized by calcifications,
valve replacement is performed. General anesthesia and cardio-pulmonary bypass
are used for all valve replacements. Most pro-cedures are performed through a
median sternotomy (ie, incision through the sternum), although the mitral valve
may be approached through a right thoracotomy incision.
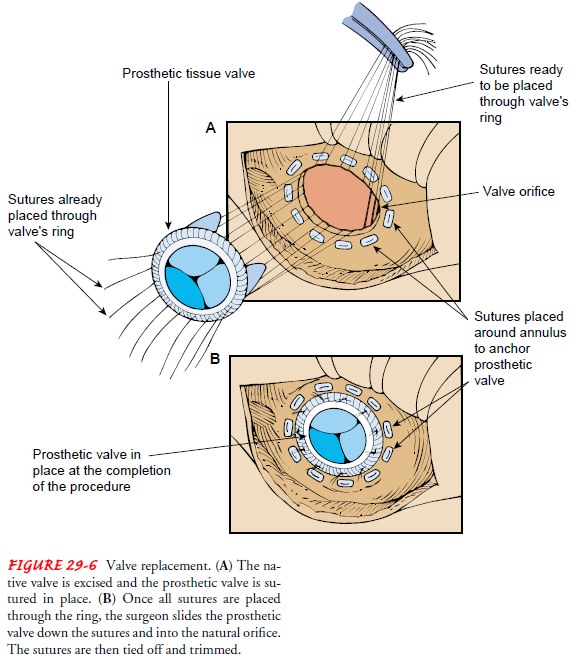
After
the valve is visualized, the leaflets and other valve struc-tures, such as the
chordae and papillary muscles, are removed. Some surgeons leave the posterior
mitral valve leaflet, its chordae, and papillary muscles in place to help
maintain the shape and function of the left ventricle after mitral valve
replacement. Su-tures are placed around the annulus and then into the valve
pros-thesis. The replacement valve is slid down the suture into position and tied
into place (Fig. 29-6). The incision is closed, and the sur-geon evaluates the
function of the heart and the quality of the prosthetic repair. The patient is
weaned from cardiopulmonary bypass, and surgery is completed.
Before surgery, the heart gradually adjusted to the pathology, but the surgery abruptly “corrects” the way blood flows through the heart. Complications unique to valve replacement are related to the sudden changes in intracardiac blood pressures. All pros-thetic valve replacements create a degree of stenosis when they are implanted in the heart. Usually, the stenosis is mild and does not effect heart function. If valve replacement was for a stenotic valve, blood flow through the heart is often improved. The signs and symptoms of the backward heart failure resolve in a few hours or days. If valve replacement was for a regurgitant valve, it may take months for the chamber into which blood had been regurgitating to achieve its optimal postoperative function.
The signs and symptoms of heart
failure resolve gradually as the heart function improves. The patient is at
risk for many postoperative compli-cations, such as bleeding, thromboembolism,
infection, conges-tive heart failure, hypertension, dysrhythmias, hemolysis,
and mechanical obstruction of the valve.
Types of Valve Prostheses
Two
types of valve prostheses may be used: mechanical and tissue (ie, biologic)
valves. Figure 29-7 shows mechanical and tissue valves.
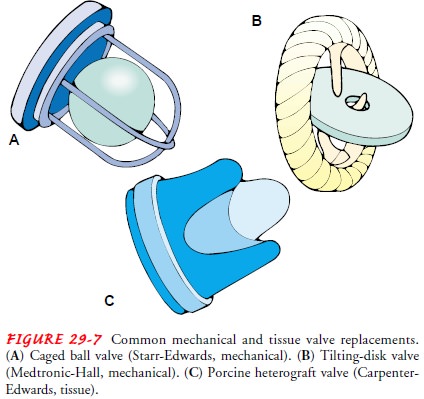
MECHANICAL VALVES
The
mechanical valves are of the ball-and-cage or disk design. Mechanical valves
are thought to be more durable than tissue prosthetic valves and often are used
for younger patients. Mechanical valves are used if the patient has renal failure,
hyper-calcemia, endocarditis, or sepsis and requires valve replacement. The
mechanical valves do not deteriorate or become infected as easily as the tissue
valves used for patients with these conditions. Thromboemboli are significant
complications associated with mechanical valves, and long-term anticoagulation
with warfarin is required.
TISSUE OR BIOLOGIC VALVES
Tissue (ie, biologic) valves are of three types: xenografts, homo-grafts, and autografts. Tissue valves are less likely to generate thromboemboli, and long-term anticoagulation is not required. Tissue valves are not as durable as mechanical valves and require replacement more frequently.
Xenografts.
Xenograftsare
tissue valves (eg, bioprostheses,het-erografts);
most are from pigs (porcine), but valves from cows(bovine) may also be used.
Their viability is 7 to 10 years. They do not generate thrombi, thereby
eliminating the need for long-term anticoagulation. They are used for women of
childbearing age because the potential complications of long-term
anticoagulation associated with menses, placental transfer to a fetus, and
delivery of a child do not exist. Xenografts also are used for patients older
than 70 years of age, patients with a history of peptic ulcer dis-ease, and
others who cannot tolerate long-term anticoagulation. Xenografts are used for
all tricuspid valve replacements.
Homografts.
Homografts,
orallografts(ie, human valves),
areobtained from cadaver tissue donations. The aortic valve and a portion of
the aorta or the pulmonic valve and a portion of the pulmonary artery are
harvested and stored cryogenically. Homo-grafts are not always available and
are very expensive. Homografts last for about 10 to 15 years, somewhat longer
than xenografts. Homografts are not thrombogenic and are resistant to subacute
bacterial endocarditis. They are used for aortic and pulmonic valve
replacement.
Autografts.
Autografts
(ie, autologous valves) are obtained byexcising the patient’s own pulmonic
valve and a portion of the pulmonary artery for use as the aortic valve.
Anticoagulation is un-necessary because the valve is the patient’s own tissue
and is not thrombogenic. The autograft is an alternative for children (it may
grow as the child grows), women of childbearing age, young adults, patients
with a history of peptic ulcer disease, and those who can-not tolerate
anticoagulation. Aortic valve autografts have remained viable for more than 20
years.
Most
aortic valve autograft procedures are double valve-replacement procedures,
because a homograft also is performed for pulmonic valve replacement. If
pulmonary vascular pressures are normal, some surgeons elect not to replace the
pulmonic valve. The patient can recover without a valve between the right
ven-tricle and the pulmonary artery.
Related Topics