Chapter: Basic Concept of Biotechnology : Biomolecules
Triacylglycerols
Triacylglycerols:
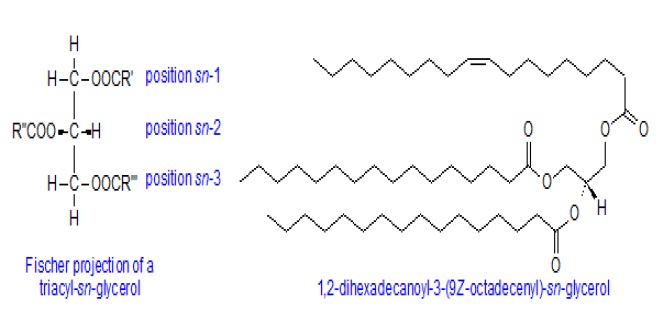
TriacylglycerolŌĆÖs (formerly called triglycerides) are complex lipids. Triacylglycerols act as energy storage molecules, especially in adipose tissue; triacylglycerols are also found in lipoproteins. Triacylglycerols are not found in membranes, because they are essentially entirely non-polar. Triacylglycerols consist of three fatty acid molecules forming ester links to glycerol. A triacylglycerol molecule can be comprised of different fatty acids, or of three identical fatty acids. In nature, they are synthesized by enzyme systems, which determine that a centre of asymmetry is created about carbon-2 of the glycerol backbone, so they exist in enantiomeric forms, i.e. with different fatty acids in each position.
A stereo specific numbering system has been recommended to describe these forms. In a Fischer projection of a natural L-glycerol derivative, the secondary hydroxyl group is shown to the left of C-2; the carbon atom above this then becomes C-1 and that below is C-3. The prefix "sn" is placed before the stem name of the compound, when the stereochemistry is defined. Their primary biological function is to serve as a store of energy. As an example, the single molecular species 1,2-dihexadecanoyl-3-(9Z-octadecenoyl)-sn-glycerol is illustrated.
Diacylglycerols (less accurately termed "diglycerides") and monoacylglycerols (monoglycerides) contain two moles and one mole of fatty acids per mole of glycerol, respectively, and exist in various isomeric forms. They are sometimes termed collectively "partial glycerides". Although they are rarely present at greater than trace levels in fresh animal and plant tissues, 1,2-diacyl-sn-glycerols are key intermediates in the biosynthesis of triacylglycerols and other lipids, and they are vital cellular messengers, generated on hydrolysis of phosphatidylinositol and related lipids by a specific phospholipase C. 2-Monoacyl-sn-glycerols are formed as intermediates or end-products of the enzymatic hydrolysis of triacylglycerols; these and other positional isomers are powerful surfactants. 2-Arachidonoylglycerol has important biological properties (as an endocannabinoid).

Acyl migration occurs rapidly in partial glycerides at room temperature, but especially on heating, in alcoholic solvents or in the presence of acid or base, so special procedures are required for their isolation or analysis if the stereochemistry is to be retained. Synthetic 1/3-monoacylglycerols are important in commerce as surfactants.
Related Topics