Chapter: Basic Concept of Biotechnology : Biomolecules
Structure of Proteins
Structure of Proteins
Protein molecules are polymers of different sizes and shapes with different physical and chemical properties. The monomer units forproteins are amino acids. Al the amino acids that are found in proteins have an amino group (-NH2) on the carbon atom adjacent to carbonyl group, hence are called α-amino acids. The general formula of α-amino acids is shown below.
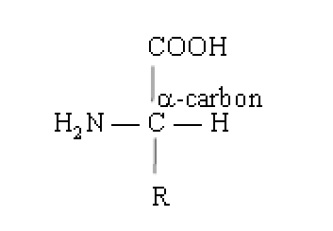
All proteins found in nature are the polymers of about twenty (20) different α-amino acids and these entire have L-configuration. Out of these ten (10) amino acids cannot be synthesized by our body and hence must form the part of our diet. These are called essential amino acids.
All proteins have one common structural feature that heir amino acids are connected to one another by peptide linkages. By a peptide
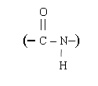 mean an amide
mean an amide
Bond formed when the carboxyl group of one amino acid molecule reacts with the- amino group of another. In the process, a molecule of water is given of. The product of the reaction is called a peptide or more precisely a dipeptide because it is made by combining two amino acids, as shown below:
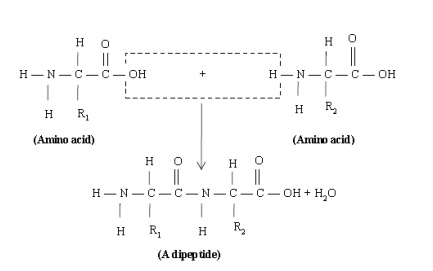
If a third amino acid is joined to a dipeptide in the same manner, the product is a tripe tide. Thus, a tripe tide contains three amino acids linked by two peptide linkages. Similar combinations of four, five, six amino acids give a tetra peptide, a pent peptide, a hexapeptide, respectively. Peptides formed by the combination of more than ten amino acid units are called polypeptides. Proteins are polypeptides formed by the combination of large number of amino acid units. There is no clear line of demarcation between polypeptides and proteins. For example insulin, although it contains only 51 amino acids, is generally considered a small protein.
The amino acid unit with the free amino group is known as the N-terminal residue and the one with the free carboxyl group is called the C-terminal residue. By convention, the structure of peptide or proteins written with the N-terminal residue on the left and the C- terminal on the right.
The actual structure of a protein can be discussed at four different levels.
Primary structure: Information regarding the sequence of aminoacids in a protein chain is called its primary structure. The primary structure of a protein determines its functions and is critical to its biological activity.
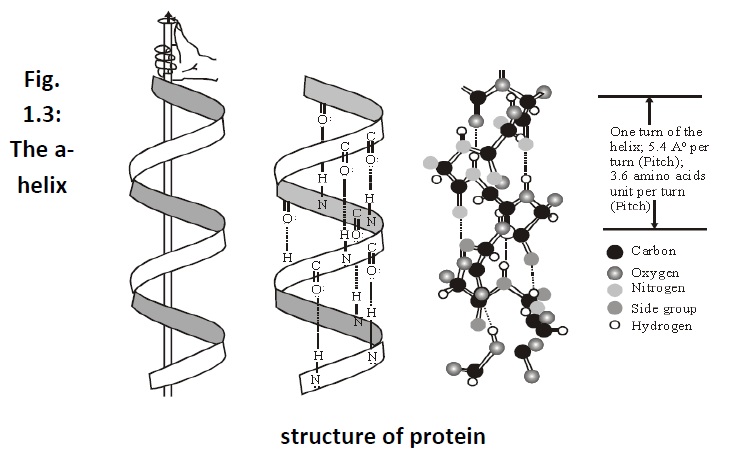
Secondary structure: The secondary structure arises due to theregular folding of the polypeptide chain due to hydrogen bonding
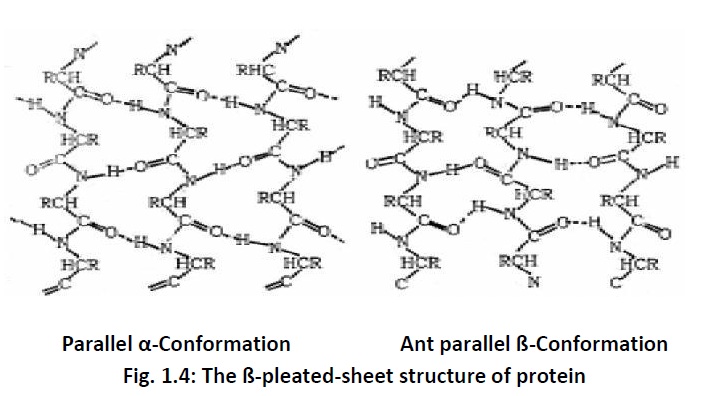
between and two types of secondary structures have been reported. These are– α helix (Fig.1.3) when the chain coils up and ß-pleated sheet (Fig. 1.4) when hydrogen bonds are formed between the chains.
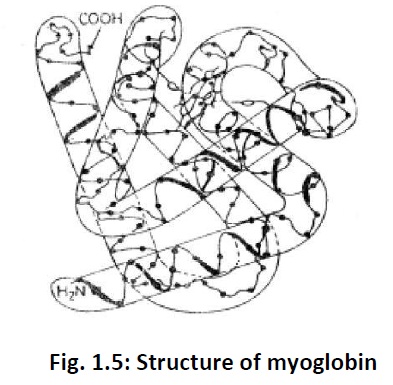
Tertiary structure: It is the three-dimensional structure of proteins. Itarises due to folding and super imposition of various α-helical chains or ß-plated sheets. For example Fig. 1.5 represents the tertiary structure for the protein myoglobin.
Quaternary structure: The quaternary structure refers to the way inwhich simple protein chains associate with each other resulting in the formation of a complex protein. By different modes of bonding in secondary and tertiary structural levels a protein molecule appears to have a unique three-dimensional structure.
Loss of Protein Structure:
Considering the small differences in the free energy of folded and unfolded proteins, it is not surprising that protein structure is especially sensitive to environmental factors. Many physical and chemical agents can disrupt a protein’s native conformation. The process of structure disruption, which may or may not involve protein unfolding, is called denaturation.
Denaturation
One of the great difficulties in the study of the structure of proteins is that if the normal environment of a living protein molecule is changed even slightly, such as by a change in pH or in temperature, the hydrogen bonds are disturbed and broken. When attractions between and within protein molecules are destroyed, the chains separate from each other, globules unfold and helices uncoil. We say that the protein has been denatured. Denaturation is seen in our daily life in many forms. The curdling of milk is caused by bacteria in the milk which produce lactic acid. The change in pH caused by the lactic acid causes denaturation, coagulation and precipitation of the milk proteins. Similarly, the boiling of an egg causes precipitation of the albumin proteins in the egg white. Some proteins (such as those in skin, fingernails, and the stomach lining) are extremely resistant to denaturation.
Related Topics