Chapter: Mechanical : Advanced IC Engines : Compression Ignition Engines
Thermodynamic analysis of CI engines
Thermodynamic analysis of CI engines:
Cylinder
pressure versus crank angle data over the compression and expansion strokes of
the engine operating cycle can be used to obtain quantitative information on
the progress of combustion. Suitable methods of analysis which yield the rate
of release of the fuel's chemical energy (often called heat release), or rate
of fuel burning, through the diesel engine combustion process will now be
described. The method of analysis starts with the first law of thermodynamics
for an open system which is quasi static (i.e., uniform in pressure and
temperature). The first law for such a system
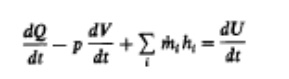
where
dQ/dt is the heat-transfer rate across the system boundary into the system,
p(dV/dt) is the rate of work transfer done by the system due to system boundaq
mass flows across the system boundary displacement, ṁ, is the mass flow rate into the system across the system boundary
at location i (flow out of the system would be negative), hi is the enthalpy of
flux i entering or leaving the
system, and U is, the energy of the material contained inside the system
boundary.
The
following problems make the application of this equation to diesel combustion
difficult:
1. Fuel is
injected into the cylinder. Liquid fuel is added to the cylinder which
vaporizes and mixes with air to produce a fuel/lair ratio distribution which is
nonunifom and varies with time. The process is not quasi static.
2. The
composition of the burned gases (also nonuniform) is not known.
3. The
accuracy of available correlations for predicting heat transfer in diesel which
cannot be determined exactly.
4. Crevice
regions (such as the volumes between the piston, rings, and cylinder wall)
constitute a few percent of the clearance volume. The gas in the regions is
cooled to close to the wall temperature, increasing its density and, therefore,
the relative importance of these crevices. Thus crevices increase heat transfer
and contain a non negligible fraction of the cylinder charge at conditions that
are different from the rest of the combustion chamber.
Due to
difficulties in dealing with these problems, both sophisticated method of
analysis and more simple methods give only approximate answers.
Related Topics