Chapter: Mechanical : Advanced IC Engines : Compression Ignition Engines
Four Stages of combustion
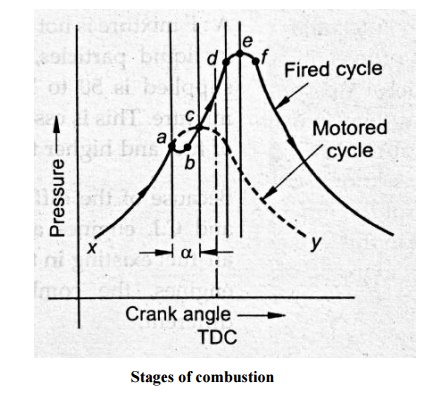
Stages of combustion:
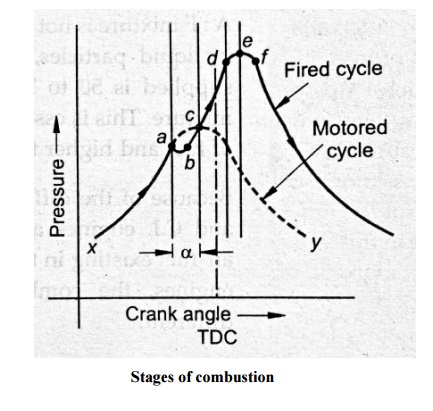
Stages of combustion
Stages of
combustion can be divided into four stages namely,
1. Pre-flame
combustion
2. Uncontrolled
combustion
3. Controlled
combustion and
4. After
burning.
Pre flame combustion:
In actual
engine cycle, the fuel injection starts at the point ‘a’ shown in fig. As soon
as the fuel jet is known into a fine spray, the fuel starts absorbing heat from
the surrounding high temperature air and vaporization of fuel starts. But in
the absence of flame, therefore it is known as pre-flame reaction. At the
beginning of pre-flame combustion, the energy release rate is very less than
rate of heat absorption by the fuel because the amount of fuel vapour is small.
As a result, the pressure in the cylinder decreases with the progressive fuel
vaporization. This decrease in pressure attains a maximum value when the energy
release due to pre-flame reaction is equal to the rate of heat absorption by
the fuel. This process of fuel vaporization and subsequent decrease in pressure
in the cylinder is shown on fig by paths
‘ab’.
As the
energy release rate due to pre-flame reaction is more than the rate of heat
absorption, the pressure inside the cylinder starts increasing. This rising
pressure intersects the pressure curve without the fuel injection at the point
‘c’. At point ‘c’, the pressure drop caused by the fuel vaporization is
completely recovered by the energy released due to preflame combustion. The
pressure inside the cylinder after the point ‘c’ rapidly increases as the
ignition takes place somewhere around the point ‘c’ and flame appears. The
actual flame (actual combustion) starts at the point ‘c’ where as the fuel
injection starts at point ‘a’. The time required to start the actual combustion
of after starting the fuel injection is known as
“delay
period” and the crank angle required for this is known as “delay period angle”
and it is shown in the fig by an angle α.
Uncontrolled Combustion:
The time
and place where ignition will stop is not fixed by anything in compression
ignition engine as in SI engines.
The air fuel
mixture in the combustion chamber before starting the combustion is very
heterogeneous and the concentration of the fuel may vary from 0 to 100%. The
first ignition (flame) generally occurs in the region of chemically correct A:F
mixture because it requires minimum reaction time. Once the ignition takes
place, the flame formed propagates through the mixture of air and vaporized
fuel and ignites the adjacent part of the charge or it may initiate the auto
ignition in the part of A:F mixture away from the flame front by transferring
the heat by radiation.
A
considerable amount of fuel is accumulated in the combustion chamber during the
relay period (time between the start of injection of fuel and start of ignition
of fuel). This accumulated fuel burns very rapidly causing a steep raise in the
cylinder pressure. The rate of pressure raise increases with the increase in
delay period because of the amount of fuel taking part in this combustion
increase with an increasing delay period. This phase of combustion causing
rapid pressure raise in the cylinder is known as “period of uncontrolled
combustion”.
Controlled combustion:
All the
accumulated fuel during the delay period generally burns during the period of
controlled combustion. The fuel injected after this (after point d) burns at
the same rate at which it is injected because, the vaporisation of fuel, mixing
with the air and burning takes place almost instantaneously as the fuel leaves
the nozzle. This is because, the temperature and pressure inside the cylinder
are sufficiently high and sufficient turbulence is created due to precious
burning, thus the delay period for the fuel injected after point “d” is almost
zero. This period of combustion is known as “controlled combustion” because the
rate of burning can be controlled by controlling the rate of injection. This is
confirmed until the supply of fuel ceases. This process is shown by the path
“de” on the fig.
After burning:
The
thermal decomposition of the part of fuel takes place during uncontrolled and
controlled combustion. The decomposed fuel molecules contain enough number of
hydrocarbons and carbon particles which has lower reaction rates. Some carbon
and hydro carbon, decomposed from fuel are left in the combustion product
because the rate of decomposition during uncontrolled and controlled combustion
is more than the rate of reaction of these molecules during that period. These
unburned hydrocarbons and carbon generally burn after stopping the fuel
injection during the expansion stroke. This process of combustion of decomposed
carbon atoms is known as “after burning”.
Related Topics