Chapter: Medical Immunology: Immunoglobulin Structure
The Antibody-Combining Site
THE ANTIBODY-COMBINING SITE
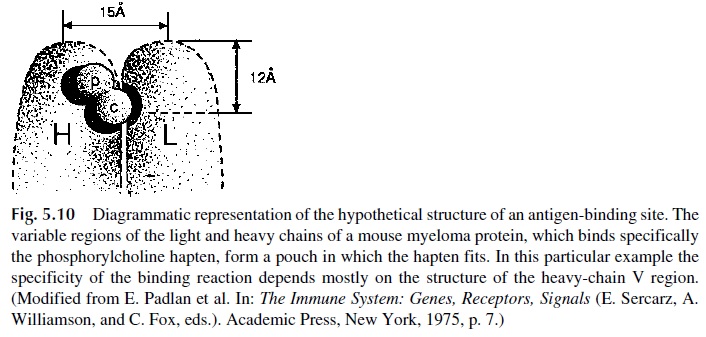
As mentioned earlier, the binding of antigens by antibody molecules takes place in the Fab region and is basically a noncovalent interaction that requires a good fit between the anti-genic determinant and the antigen-binding site on the immunoglobulin molecule. The anti-gen-binding site appears to be formed by the variable regions of both heavy and light chains, folded in close proximity forming a pouch where an antigenic determinant or epi-tope will fit (Fig. 5.10).
Actually, certain sequence stretches of the variable regions vary widely from protein to protein, even among proteins sharing the same type of variable regions. For this reason, these highly variable stretches have been designated as hypervariable regions. The struc-ture of hypervariable regions is believed to play a critical role in determining antibody specificity since these regions are believed to be folded in such a way that they form a “pouch” where a given epitope of an antigen will fit. In other words, the hypervariable re-gions will interact to create a binding site whose configuration is complementary to that of a given epitope. Thus, these regions can be also designated as complementarity-determin-ing regions.
Related Topics