Chapter: Genetics and Molecular Biology: Generating Genetic Diversity: Antibodies
Telling the Difference Between Foreign and Self
Telling the Difference Between Foreign and Self
What keeps a vertebrate from synthesizing
antibodies against its own macromolecules? In principle, one solution to this
problem would be to have the entire repertoire of an animal’s antibody
specificities explicitly encoded in the genome and expressed during
development. Then the problem of preventing synthesis of antibodies against
itself would be simple. None would be coded in the collection of specificities.
This would be inefficient for at least two reasons. First, as we will see in
the next section, the very large number of specificities needed by the immune
system would use an inordinate amount of the coding potential of the genome.
Second, such an inflexible system would be highly dangerous, for the moment an
invader mutated and developed surface molecules not neutralized by the host
cells’ immune system, the invader could freely grow in the host animal, and the
entire population of host animals could be killed. It would be as though the
animals didn’t possess any immune system at all, for the mutated invader would
encounter no opposition.
For these reasons we expect considerable
randomness, even in ge-netically identical animals, in the generation of
antibody-binding spe-cificities. Therefore, at least a few animals in any
population will be capable of recognizing any antigen. It is this randomness,
however, that generates the problem of responding only to foreign
macromolecules. If the generation of the ability to recognize diverse antigens
is random and not explicitly encoded in the DNA, the same randomness will
generate antibodies against the organism itself, and these self-recogniz-ing
antibodies must be eliminated if the animal is to survive.
The problem of preventing self-recognition is
largely solved by killing the lymphocytes that recognize macromolecules of the
organism itself. This, of course, must be done after development of the immune
system but before the organism is exposed to foreign macromolecules. For
purposes of the molecular biology of the immune system, it is useful to
consider the following simplified picture of the development of an
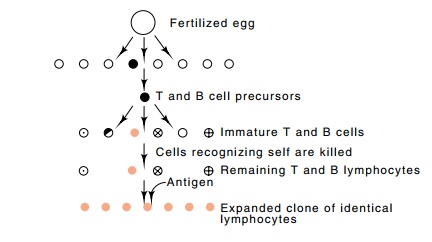
Figure
20.1 Growth and differentiation of a
fertilized egg to yield the immature T cells, some of which recognize self and
are killed. The remainder recognize other antigens and reside in the animal
until the binding of antigen to their surface triggers them to begin growth,
after which "clonal expansion" yields a population of size sufficient
to synthesize appreciable quantities of immunoglobulin.
animal’s lymphocytes (Fig. 20.1). Eventually in the
cells deriving from a fertilized egg, there arises a single cell that can be
considered the precursor of the T and B cells. Subsequent divisions of this
cell produce only T and B cells. Each of the T or B cells generally develops
the ability to recognize only one antigen. Some of these antigens are
macromole-cules present in the animal itself. The lymphocytes that recognize
these “self” antigens are either killed or made unresponsive. The time of such
deletion or inactivation varies with the animal. In mice this occurs at about
the time of birth, whereas in humans it is much earlier. The lymphocytes
capable of synthesizing antibodies to antigens not present at this time survive
and form the pool of immature T and B lymphocytes that are capable of
responding to foreign antigens.
The consequences of incomplete killing of
lymphocytes that recog-nize “self” or the subsequent alteration of specificity
so that “self” is recognized are predictable. In either case the immune system
turns against the animal, and autoimmune conditions result. Autoimmunity is not
at all uncommon. Forms of diabetes mellitus result from improper recognition of
pancreatic cells. Myasthenia gravis is a condition in which antibodies are
directed against muscle-nerve connections, and systemic lupus erythematosus is
a disease in which antibodies are directed against many cellular components.
Many other examples are also known. Autoimmune conditions also appear to result
from loss of the ability to repress activation of T or B cells.
Related Topics