Chapter: Medical Physiology: Rhythmical Excitation of the Heart
Specialized Excitatory and Conductive System of the Heart
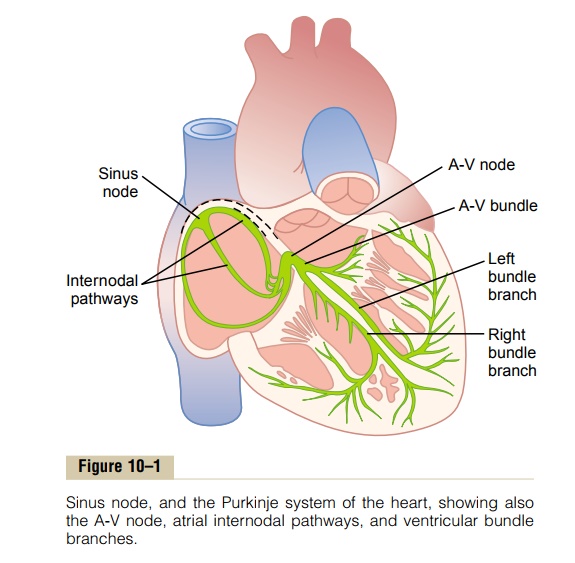
Specialized Excitatory and Conductive System of the Heart
Figure 10–1 shows the specialized excitatory and conductive system of the heart that controls cardiac contractions. The figure shows the sinus node(also called sinoatrial or S-A node), in which the normal rhythmical impulse is generated;the internodal pathways that conduct the impulse from the sinus node to the atrioventricular (A-V) node; the A-V node, in which the impulse from the atria is delayed before passing into the ventricles; the A-V bundle, which conducts the impulse from the atria into the ventricles; and the left and right bundlebranches of Purkinje fibers, which conduct the cardiac impulse to all parts ofthe ventricles.
Sinus (Sinoatrial) Node
The sinus node (also called sinoatrial node) is a small, flattened, ellipsoid strip of specialized cardiac muscle about 3 millimeters wide, 15 millimeters long, and 1 millimeter thick. It is located in the superior posterolateral wall of the right atrium immediately below and slightly lateral to the opening of the superior vena cava. The fibers of this node have almost no contractile muscle filaments and are each only 3 to 5 micrometers in diameter, in contrast to a diameter of 10 to 15 micrometers for the surrounding atrial muscle fibers. However, the sinus nodal fibers connect directly with the atrial muscle fibers, so that any action potential that begins in the sinus node spreads immediately into the atrial muscle wall.
Automatic Electrical Rhythmicity of the Sinus Fibers
Some cardiac fibers have the capability of self-excitation, a process that can cause automatic rhythmical discharge and contraction. This is especially true of the fibers of the heart’s specialized conducting system, including the fibers of the sinus node. For this reason, the sinus node ordinarily controls the rate of beat of the entire heart. First, let us describe this automatic rhythmicity.
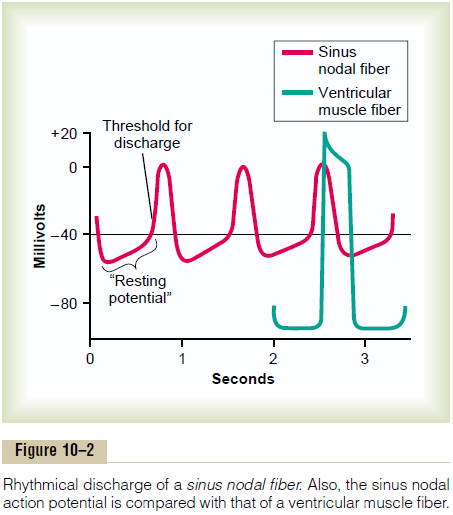
Mechanism of Sinus Nodal Rhythmicity. Figure 10–2 showsaction potentials recorded from inside a sinus nodal fiber for three heartbeats and, by comparison, a single ventricular muscle fiber action potential. Note that the “resting membrane potential” of the sinus nodal fiber between discharges has a negativity of about -55 to -60 millivolts, in comparison with -85 to -90 millivolts for the ventricular muscle fiber. The cause of this lesser negativity is that the cell membranes of the sinus fibers are naturally leaky to sodium and calcium ions, and positive charges of the entering sodium and calcium ions neutralize much of the intracellular negativity.
Before attempting to explain the rhythmicity of the sinus nodal fibers, first recall from the discussions that cardiac muscle has three types of membrane ion channels that play important roles in causing the voltage changes of the action potential. They are (1) fast sodium channels, (2) slow sodium-calcium channels, and (3) potassium channels. Openingof the fast sodium channels for a few 10,000ths of a second is responsible for the rapid upstroke spike of the action potential observed in ventricular muscle, because of rapid influx of positive sodium ions to the interior of the fiber. Then the “plateau” of the ven-tricular action potential is caused primarily by slower opening of the slow sodium-calcium channels, which lasts for about 0.3 second. Finally, opening of potas-sium channels allows diffusion of large amounts of positive potassium ions in the outward direction through the fiber membrane and returns the mem-brane potential to its resting level.
But there is a difference in the function of these channels in the sinus nodal fiber because the “resting” potential is much less negative—only -55 millivolts in the nodal fiber instead of the -90 millivolts in the ven-tricular muscle fiber. At this level of -55 millivolts, the fast sodium channels mainly have already become “inactivated,” which means that they have become blocked. The cause of this is that any time the mem-brane potential remains less negative than about -55 millivolts for more than a few milliseconds, the inacti-vation gates on the inside of the cell membrane that close the fast sodium channels become closed and remain so. Therefore, only the slow sodium-calcium channels can open (i.e., can become “activated”) and thereby cause the action potential. As a result, the atrial nodal action potential is slower to develop than the action potential of the ventricular muscle. Also, after the action potential does occur, return of the potential to its negative state occurs slowly as well, rather than the abrupt return that occurs for the ventricular fiber.
Self-Excitation of Sinus Nodal Fibers. Because of thehigh sodium ion concentration in the extracellular fluid outside the nodal fiber, as well as a moderate number of already open sodium channels, positive sodium ions from outside the fibers normally tend to leak to the inside. Therefore, between heartbeats, influx of positively charged sodium ions causes a slow rise in the resting membrane potential in the positive direction. Thus, as shown in Figure 10–2, the “resting” potential gradually rises between each two heartbeats. When the potential reaches a threshold voltage of about -40 millivolts, the sodium-calcium channels become “activated,” thus causing the action potential. Therefore, basically, the inherent leakiness of the sinus nodal fibers to sodium and calcium ions causes their self-excitation.
Why does this leakiness to sodium and calcium ions not cause the sinus nodal fibers to remain depolarized all the time? The answer is that two events occur during the course of the action potential to prevent this. First, the sodium-calcium channels become inac-tivated (i.e., they close) within about 100 to 150 mil-liseconds after opening, and second, at about the same time, greatly increased numbers of potassium channels open. Therefore, influx of positive calcium and sodium ions through the sodium-calcium channels ceases, while at the same time large quantities of positive potassium ions diffuse out of the fiber. Both of these effects reduce the intracellular potential back to its negative resting level and therefore terminate the action potential. Furthermore, the potassium channels remain open for another few tenths of a second, tem-porarily continuing movement of positive charges out of the cell, with resultant excess negativity inside the fiber; this is called hyperpolarization. The hyperpolar-ization state initially carries the “resting” membrane potential down to about -55 to -60 millivolts at the termination of the action potential.
Last, we must explain why this new state of hyper-polarization is not maintained forever. The reason is that during the next few tenths of a second after the action potential is over, progressively more and more potassium channels close. The inward-leaking sodium and calcium ions once again overbalance the outward flux of potassium ions, and this causes the “resting” potential to drift upward once more, finally reaching the threshold level for discharge at a potential of about -40 millivolts. Then the entire process begins again: self-excitation to cause the action potential, recovery from the action potential, hyperpolarization after the action potential is over, drift of the “resting” potential to threshold, and finally re-excitation to elicit another cycle. This process continues indefinitely throughout a person’s life.
Internodal Pathways and Transmission of the Cardiac Impulse Through the Atria
The ends of the sinus nodal fibers connect directly with surrounding atrial muscle fibers. Therefore, action potentials originating in the sinus node travel outward into these atrial muscle fibers. In this way, the action potential spreads through the entire atrial muscle mass and, eventually, to the A-V node. The velocity of con-duction in most atrial muscle is about 0.3 m/sec, but conduction is more rapid, about 1 m/sec, in several small bands of atrial fibers.
One of these, called the anterior interatrial band, passes through the anteriorwalls of the atria to the left atrium. In addition, three other small bands curve through the anterior, lateral, and posterior atrial walls and terminate in the A-V node; shown in Figures 10–1 and 10–3, these are called, respectively, the anterior, middle, and posterior inter-nodal pathways. The cause of more rapid velocity ofconduction in these bands is the presence of special-ized conduction fibers. These fibers are similar to even more rapidly conducting “Purkinje fibers” of the ven-tricles, which will be discussed.
Atrioventricular Node, and Delay of Impulse Conduction from the Atria to the Ventricles
The atrial conductive system is organized so that the cardiac impulse does not travel from the atria into the ventricles too rapidly; this delay allows time for the atria to empty their blood into the ventricles before ventricular contraction begins. It is primarily the A-Vnode and its adjacent conductive fibersthat delay thistransmission into the ventricles.
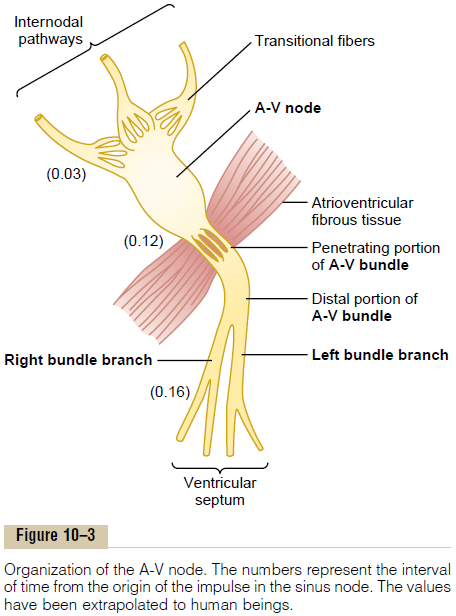
The A-V node is located in the posterior wall of the right atrium immediately behind the tricuspid valve, as shown in Figure 10–1. And Figure 10–3 shows dia-grammatically the different parts of this node, plus its connections with the entering atrial internodalpathway fibers and the exiting A-V bundle. The figurealso shows the approximate intervals of time in fractions of a second between initial onset of the cardiac impulse in the sinus node and its subsequent appearance in the A-V nodal system. Note that the impulse, after traveling through the internodal path-ways, reaches the A-V node about 0.03 second after its origin in the sinus node. Then there is a delay of another 0.09 second in the A-V node itself before the impulse enters thepenetrating portion of the A-Vbundle, where it passes into the ventricles.A final delayof another 0.04 second occurs mainly in this penetrat-ing A-V bundle, which is composed of multiple small fascicles passing through the fibrous tissue separating the atria from the ventricles.
Thus, the total delay in the A-V nodal and A-V bundle system is about 0.13 second. This, in addition to the initial conduction delay of 0.03 second from the sinus node to the A-V node, makes a total delay of 0.16 second before the excitatory signal finally reaches the contracting muscle of the ventricles.
Cause of the Slow Conduction. The slow conduction in thetransitional, nodal, and penetrating A-V bundle fibers is caused mainly by diminished numbers of gap junc-tions between successive cells in the conducting path-ways, so that there is great resistance to conduction of excitatory ions from one conducting fiber to the next. Therefore, it is easy to see why each succeeding cell is slow to be excited.
Rapid Transmission in the Ventricular Purkinje System
Special Purkinje fibers lead from the A-V node through the A-V bundle into the ventricles. Except for the initial portion of these fibers where they penetrate the A-V fibrous barrier, they have functional charac-teristics that are quite the opposite of those of the A-V nodal fibers. They are very large fibers, even larger than the normal ventricular muscle fibers, and they transmit action potentials at a velocity of 1.5 to 4.0 m/sec, a velocity about 6 times that in the usual ventricular muscle and 150 times that in some of the A-V nodal fibers. This allows almost instantaneous transmission of the cardiac impulse throughout the entire remainder of the ventricular muscle.
The rapid transmission of action potentials by Purk-inje fibers is believed to be caused by a very high level of permeability of the gap junctions at the intercalated discs between the successive cells that make up the Purkinje fibers. Therefore, ions are transmitted easily from one cell to the next, thus enhancing the velocity of transmission. The Purkinje fibers also have very few myofibrils, which means that they contract little or not at all during the course of impulse transmission.
One-Way Conduction Through the A-V Bundle. A specialcharacteristic of the A-V bundle is the inability, except in abnormal states, of action potentials to travel back-ward from the ventricles to the atria. This prevents re-entry of cardiac impulses by this route from the ventricles to the atria, allowing only forward conduc-tion from the atria to the ventricles.
Furthermore, it should be recalled that everywhere, except at the A-V bundle, the atrial muscle is sepa-rated from the ventricular muscle by a continuous fibrous barrier, a portion of which is shown in Figure 10–3. This barrier normally acts as an insulator to prevent passage of the cardiac impulse between atrial and ventricular muscle through any other route besides forward conduction through the A-V bundle itself. (In rare instances, an abnormal muscle bridge does penetrate the fibrous barrier elsewhere besides at the A-V bundle. Under such conditions, the cardiac impulse can re-enter the atria from the ventricles and cause a serious cardiac arrhythmia.)
Distribution of the Purkinje Fibers in the Ventricles—The Left and Right Bundle Branches. After penetrating the fibroustissue between the atrial and ventricular muscle, the distal portion of the A-V bundle passes downward in the ventricular septum for 5 to 15 millimeters toward the apex of the heart, as shown in Figures 10–1 and 10–3. Then the bundle divides into left and right bundlebranches that lie beneath the endocardium on the tworespective sides of the ventricular septum. Each branch spreads downward toward the apex of the ven-tricle, progressively dividing into smaller branches. These branches in turn course sidewise around each ventricular chamber and back toward the base of the heart. The ends of the Purkinje fibers penetrate about one third the way into the muscle mass and finally become continuous with the cardiac muscle fibers.
From the time the cardiac impulse enters the bundle branches in the ventricular septum until it reaches the terminations of the Purkinje fibers, the total elapsed time averages only 0.03 second. Therefore, once the cardiac impulse enters the ventricular Purkinje con-ductive system, it spreads almost immediately to the entire ventricular muscle mass.
Transmission of the Cardiac Impulse in the Ventricular Muscle
Once the impulse reaches the ends of the Purkinje fibers, it is transmitted through the ventricular muscle mass by the ventricular muscle fibers themselves. The velocity of transmission is now only 0.3 to 0.5 m/sec, one sixth that in the Purkinje fibers.
The cardiac muscle wraps around the heart in a double spiral, with fibrous septa between the spiraling layers; therefore, the cardiac impulse does not neces-sarily travel directly outward toward the surface of the heart but instead angulates toward the surface along the directions of the spirals. Because of this, transmis-sion from the endocardial surface to the epicardial surface of the ventricle requires as much as another 0.03 second, approximately equal to the time required for transmission through the entire ventricular portion of the Purkinje system. Thus, the total time for trans-mission of the cardiac impulse from the initial bundle branches to the last of the ventricular muscle fibers in the normal heart is about 0.06 second.
Summary of the Spread of the Cardiac Impulse Through the Heart
Figure 10–4 shows in summary form the transmission of the cardiac impulse through the human heart. The numbers on the figure represent the intervals of time, in fractions of a second, that lapse between the origin of the cardiac impulse in the sinus node and its appear-ance at each respective point in the heart. Note that the impulse spreads at moderate velocity through the atria but is delayed more than 0.1 second in the A-V nodal region before appearing in the ventricular septal A-V bundle. Once it has entered this bundle, it spreads very rapidly through the Purkinje fibers to the entire endocardial surfaces of the ventricles.Then the impulse once again spreads slightly less rapidly through the ventricular muscle to the epicardial surfaces.
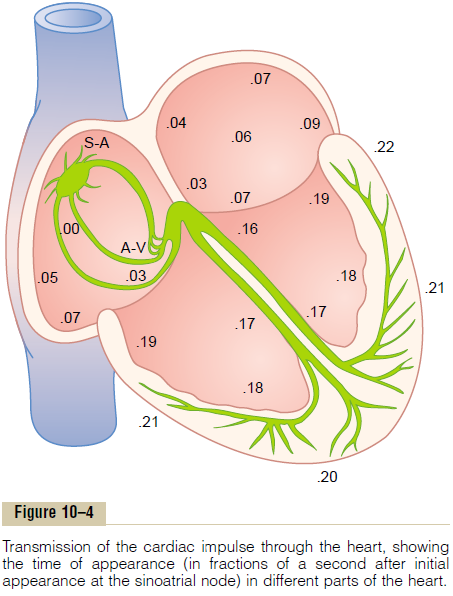
It is extremely important that the student learn in detail the course of the cardiac impulse through the heart and the precise times of its appearance in each separate part of the heart, because a thorough quanti-tative knowledge of this process is essential to the understanding of electrocardiography.
Related Topics