Chapter: Aquaculture Principles and Practices: Tilapias
Spawning and production of seed stock - Tilapias
Spawning and production of seed stock
It is somewhat paradoxical that mass production of seed stock of a group of species that spawns easily, early and frequently should present problems. But in point of fact, one of the major constraints to large-scale commercial tilapia farming is the scarcity of fry. Some of the species attain maturity as early as three months old, and under favourable temperature conditions breed in successive cycles at four to six week intervals. This results in continuous production of fry, but the farmer finds it difficult to obtain sufficient fry of the required size at any particular time. The traditional practice of harvesting tilapia ponds at regular intervals during the culture period, to remove larger fish and to allow the fry and young fish (produced by wild spawning) to reach market size before the next harvest, only results in lengthening the culture period and allowing heavy wild spawning in the ponds. This system also has the disadvantage that a good proportion of the unharvested stock would be individuals selected for slow growth. When used for further fry production, there is a likelihood of this characteristic of slow growth being passed on genetically to the offspring.
In sub-tropical or temperate regions, spawning may be limited to the summer. Even here, depending on the preferred spawning temperature range (usually 20–30°C), the fish would spawn several times and overpopulate ponds with fry of different sizes. Thus, the problem of overpopulation and stunting applies also to these regions, though not to the same extent as in the tropics.
Methods of spawning
Attempts have been made to develop a suitable fry production system for tilapias. Campbell (1978) described a relatively simple method of producing large numbers of T.nilotica fry using 600 m2 earth ponds of about 0.4m depth. Female fish of about 700 g weight and males of 200 g are stocked in one pond at an average density of one per 2 m2 in the sex ratio of one male to four or five females. They are fed on a high-protein diet for about a month, by which time they will have started spawning. The brood fish are then transferred to a second pond, where they are fed in the same way as in the first pond. Feeding is continued in the first pond as well for another month, by the end of which the fry will have reached a size of about 4cm and on average about 5000 fry are available for harvest. By this time, spawning will have occurred in the second pond and the brood fish can be transferred back to the first pond for further spawning. The production per month by this method is reported to be about 4.2 fry per m2 or 10.4 fry per female.
In Israel, ponds ranging in size from a few square metres to 5 ha, with gently sloping bottoms, are used for spawning T. nilotica and T. aurea. The ponds are dried prior to spawningto eradicate weed-fish and pests. They are filled to a depth of 50–60 cm, which is the preferred depth for spawning of these species. As the number of eggs per spawning depends on the size of the females, the stocking rate is varied according to their size. While a 100 g T. nilotica spawns about 100 eggs, a 600–1000 g fish will spawn about 1000–1500 eggs. A female T. aurea of about 1000 g weight may spawn about 2000 eggs each time. The stocking rate for males is generally 100–250 per ha.
In the Philippines, land-based spawning ponds as well as open-water-based cages or hapas are used for spawning and fry rearing. Many farms use hapas made of nylon mosquito netting to breed T. nilotica and hybrids of T. nilotica x T. mossambica. The brood fish are maintained in hapas installed in ponds with about 1 m depth of water. The fish continue to breed throughout the year. A 1 : 3 male to female sex ratio has been found to be suitable. The fry are collected at intervals of about a month and grown to fingerling stage in nursery ponds or cloth tanks. For cross-breeding, the best sex ratio has been found to be one male to three females. The average monthly production in computed to be about 1466/m3.
Open-water-based cage hatcheries used in the Philippines consist of double-walled net cages very much like the double-walled hapas used for carp hatching in India. The inner coarse-mesh (30 mm) net measures 10 x 2 x 1 m, and the outer fine-mesh net, 12 x 4 x 1.5 m. They are installed in protected calm areas of lakes, such as the ones found in Laguna de Bay. Breeders are stocked at a density of four per m2 with the same 1 : 3 sex ratio as in ponds, and fed with fine rice bran at 3 per cent body weight per day. Spawning occurs at regular intervals and the fry are collected and stocked in rearing hapas (10 x 2 x 1.5 m) at the rate of 1000 per m2. Fine rice bran is used for feeding the fry at the rate of about 6–8 per cent of body weight. After two weeks of rearing in hapas, the fingerlings are transferred to larger-meshed (6.5 mm) cages at the rate of 250–500 per m2 and fed with fine rice bran at the rate of 4–6 per cent of body weight per day.
Systems that allow a high degree of environmental control make year-round spawning of tilapias possible in temperate climates. Removal of eggs from incubating females and hatching and rearing them separately in special containers helps to increase spawning frequency and thereby overall fry production. Another important advantage of spawning under controlled conditions is that genetic purity of lines can be maintained, and this is of special importance in hybrid production. The sex ratio of females to males is generally 3: 1 or 4 : 1. As tilapia spawn at frequent intervals, harvesting has to be carried out every fortnight, when the fry are about 0.5 g in weight.
The aggressive behaviour of the male in an aquarium or tank manifests when mature fish are introduced at a size of about 100 g. Long aquarium tanks (200 x 50 x 40 cm) are stocked with immature, four-to-five-month-old fish. One male and seven to ten females form a ‘family’ in each aquarium. When they become sexually mature, the males of mouth-brooding tilapia species dig nests at the bottom if there is sand or gravel there. Even if the bottom is bare they exhibit digging movements. The male chooses the ripest female and, after a period of courtship which may last several days, spawning and fertilization take place in the nest or the bottom of the aquarium. Soon after, the female picks up the eggs in her mouth. The male then
chooses another ripe female for courtship and spawning. The eggs are removed from the female’s mouth after three to five days for further incubation. This helps in preventing cannibalism and early preparation of the female for further spawning. Zuge jars or containers placed on a shaking platform (for keeping the eggs separate and in continuous movement) are used in incubation. The eggs hatch out in about 50 hours at temperatures of 25–27°C. The larvae remain in the incubating containers until the yolk sacs are absorbed, which may take about 8–10 days.
Nursing of normal fry or mono-sex hybrids is carried out in nursery or rearing ponds. Stocking densities vary from 50 000 to 100 000 per ha, depending on the size of fingerlings to be raised. When manual sexing of fry is needed, it is necessary to grow them to a size of at least 20–50 g so as to distinguish secondary sexual characteristics with ease. Even a larger size of 100 g is often recommended, but it has to be ensured that the stock are removed before they reach maturity.
Mono-sex seed stock and hybrids
One of the methods in controlling wild spawning of tilapias is mono-sex culture, and since the male tilapia grows faster and attains a larger size, interest has been focused on producing all-male seed stock. Obviously the simplest means is to sort out males from unsorted stocks of fry, commonly known among fish culturists as hand sexing (or manual sexing). The sexes can be distinguished by visual examination of the urino-genital papillae. In the females, the papilla has two orifices whereas the male has only one. Often the female has a smaller genital papilla. This requires some skill and carefulness and can be done reliably only with fingerlings of 20–50 g size. Even at this size, there is likely to be a certain percentage of error in sorting, and even a small number of females in the stock can initiate wild spawning in production ponds. It also involves waste of female fish, although some farmers use the sorted females for preparing feeds for males to be grown to market size. Despite the skilled manual labour involved, sorting of males for commercial scale culture is practised in a number of countries. Growth and production are substantially increased, even though some wild spawning does take place in the ponds. Draining of ponds after harvesting makes it possible to start the next crop with sorted seed stock.
Another approach to the production of mono-sex stock has been by the use of steroid hormones to achieve sex reversal. As mentioned, it has been possible to reverse the sex of genotypic females by the administration of methyltestosterone or ethinyltestosterone in T. mossambica, T. nilotica and T. aurea (Guerrero, 1982). Thedegree of success varied between 90 and 100 per cent. The steroids were incorporated in the feed of fry at rates ranging from 10 to 60mg per kg fish, for durations varying from 18 to 60 days. Feminization of genotypic males was achieved in 90–100 per cent of the males of the above mentioned species, by the administration of oestrogens (ethinyloestradiol, oestrone and diethylstilboestrol). The dose consisted of 50–100mg/kg of ethinyloestradiol (in experiments with T. aurea a 100mg/kg dose of methallubure was included), 200 mg/kg of oestrone and 100mg/kg diethylstilboestrol and the duration of treatment varied between 19 and 56 days. From comparative experiments, Hanson et al. (1983) found that the sex-reversed malepopulations have a higher growth rate than hybrids and females.
Rothbard et al. (1983) have described the procedure adopted in Israel to produce hormonally sex-inverted all-male tilapia. The fry are placed in outdoor concrete circular tanks of 28 m3 capacity (diameter 6 m). Commercial high-protein trout starter feed or eel feed is mixed with the androgen 17 a-ethinyltestos-terone dissolved in 95 per cent ethanol (technical grade) for feeding the fry. The ethanol is evaporated by drying the mixture in the sun for several hours and the fry are fed at the rate of 12 per cent of their body weight per day. The tanks are protected from sunlight and the water temperature maintained between 21 and 22.5°C. The treatment lasts about 28–29 days.
Treatment of hybrids of T. nilotica males and T. aurea females, F-1 hybrids of T. nilotica and T. aurea, and the red tilapia yield populationscontaining 98–100 per cent males. Studies showed the level of testosterone in the plasma of sex-inverted fish to be only 11.1 ± 4.3 ng/ml, compared to sexually active males of T. nilotica and T.hornorum with 37.8 ± 9.1 ng/ml and 41.7 ± 4.6 ng/ml respectively. From this it is concluded that androgen treatment of fry has no effect on circulating testosterone levels at post-maturation (Rothbard et al., 1982).
Hopes of using inter-specific hybrids as a means of controlling wild spawning were aroused by the production of all-male progeny by crossing T. mossambica females withT. hornorum males by Hickling (1960). Besidesproducing mono-sex male populations, cross-breeding could help in improving catchability, growth rate, temperature tolerance and body coloration. These could greatly enhance the value of tilapia as candidate species for large-scale fish culture. Since then a number of all-male or predominantly male hybrids have been produced:
T. nilotica x T. hornorum (Pruginin andKanyike, 1965)
T. nilotica x T. aurea (Fishelson, 1962)
T. nilotica x T. variabilis (Pruginin, 1967)
T. spilurus niger x T. hornorum (Pruginin, 1967)
T. vulcani x T. hornorum (Pruginin, 1967)
T. vulcani x T. aurea (Pruginin, 1967)
T. nilotica x T. macrochir (Lessent, 1968)
One hybrid that has received special attention from fish culturists for some time is the so-called red tilapia, the colour of which is a blend of pink, yellow and gold. It is appreciated in the market in preference to the normally silvery grey or black-coloured tilapia. Red tilapia is known to have a faster growth rate and food conversion ratio. It can grow in both fresh- and brackish-water environments. The origin of this hybrid is not yet fully documented. A reddish-orange F-2 progeny with superior qualities was obtained in Taiwan by crossing a mutant reddish-orange female of T. mossambica with a normal-coloured grey male T. nilotica. In the Philippines, a similar reddish-orange or golden progeny was obtained by cross-breeding a female hybrid of T. mossambica x T. hornorum with a strain of T. nilotica. German and Avatlion (1983) found that the red tilapia is intermediate in several characteristics between T. mossambica, T. hornorum, T. nilotica and T. aurea, and speculated that all these species areinvolved in the hybrid.
Lovshin (1982), who has reviewed experience in tilapia hybridization, pointed out that in spite of the knowledge that all-male or pre-dominantly male populations can be produced by hybridization, commercial culture of such hybrids is limited. One reason for this is the difficulty in maintaining pure genetic lines which are necessary to obtain consistent results in hybridization. In commercial production, varying proportions of females occur as a result of contamination of the brood-stock lines. Electrophoretic comparisons of blood proteins and crossing in aquaria of the brood stock until all-male offspring are consistently produced have been suggested as a means of ensuring pure brood stocks. These procedures are feasible in breeding centres, but there are only a few countries where such facilities are available at present for the production and distribution of selected pure lines of aquaculture species. Further investigations by Majumdar and McAndrew (1983) showed that even crosses between pure lines produce varying sex ratios. In 41 trials, only one cross (T. mossambica malesx T. macrochir females gave 100 percent male progeny.
Small aquaria, concrete tanks and plastic pools are generally used for hybrid production. As mentioned earlier, male aggression is a problem in spawning operations in small containers. Cannibalism of fingerlings on newly hatched larvae is an additional problem in aquaria and similar containers. According to Lovshin (1982), some hybrid crosses are difficult to carry out in small confined environments. It has been demonstrated that earthen ponds can be successfully used in cross-breeding of tilapias, following the general procedures for pond spawning.
Lovshin (1982) has described a system developed in Brazil for the production of hybrids of T. hornorum and T. nilotica. Fig. 20.5 illustratesthe steps recommended. Fingerlings of the two species are sexed when they have reached weights of 20–30 g, and the males and females are stocked separately in segregated brood-stock preparation ponds, at the rate of two or three per m2. They are fed at the rate of 5 per cent of their body weight daily, and in two to
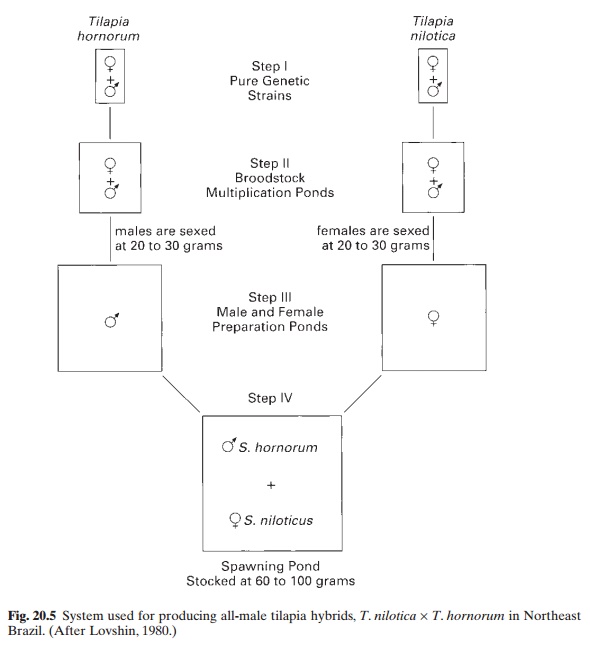
Mature male T. hornorum and female T. nilotica which have swollen genital papillae areintroduced in the spawning pond at a ratio of 1 : 1. The stocking rate is one female to every 7 m2 pond surface. The brood stock are fed with agricultural byproducts at the same rate as for immature fish. After about two and a half months, the spawning ponds are drained and the hybrid fingerlings collected for rearing in nursery ponds. The ponds are dried after drain ing and poisoned to eliminate any small fry that may have been produced by back-crossing between all-male hybrids and female T. nilotica. The process can then be repeated withbrood stock that have produced sufficient numbers of hybrid fingerlings.
According to Hepher and Pruginin (1981), tilapias do not readily hybridize in aquaria and so artificial propagation methods are generally used for hybrid production in Israel. Ripe spawners with swollen papilla and intense pigmentation are selected. The eggs from one of the females of the selected species are stripped into a suitable container and mixed for about two minutes with milt from the male of the other species. About 10 ml saline solution is added and the eggs stirred for a further two minutes. The eggs can then be rinsed and transferred to an incubator for hatching.
Related Topics