Chapter: Clinical Dermatology: The function and structure of the skin
Some cellular components of the skin immune system
Some
cellular components of the skin immune system
Keratinocytes
Their
prime role is to make the protective horny layer and to support to the outermost epithelium of
the body but they also have immunological func-tions in their own right.
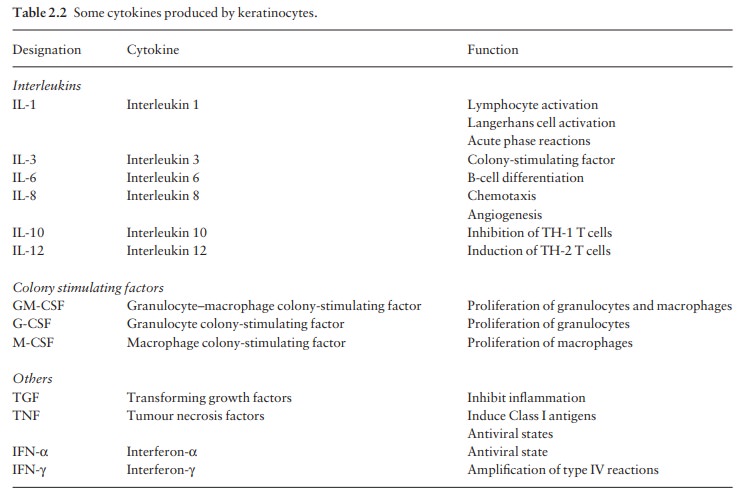
Keratinocytes produce large numbers of cytokines (see Table 2.2), and can be
induced by γ-interferon
to express HLA-DR. They can also produce α-melanocyte-stimulating
hormone, which is immunosuppressive. Keratinocytes play a central part in
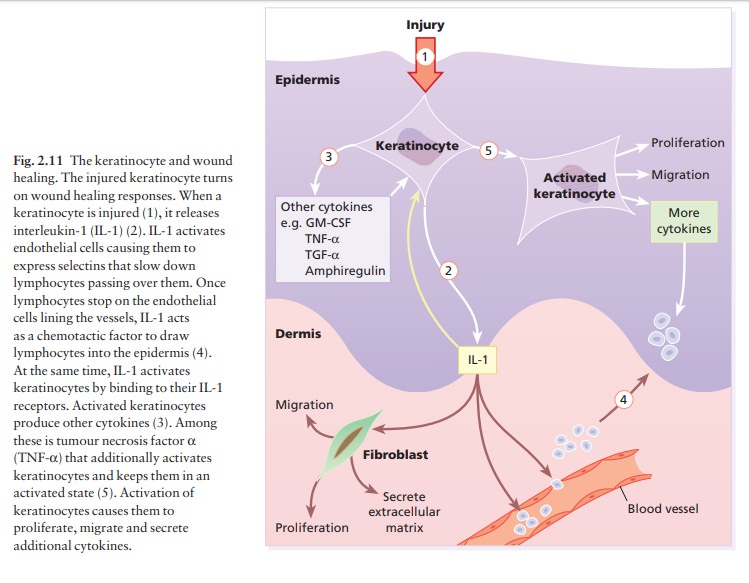
healing after epidermal injury (Fig. 2.11).
Langerhans cells
These
dendritic cells come from the bone marrow and circulate through the epidermis,
the dermis, lymphatics (as ‘veiled cells’), and also through the T-cell area of
the lymph nodes where they are called ‘dendritic’ or ‘interdigitating’ cells.
They can be identified in tissue sections by demonstrating their characteristic
surface markers (e.g. CD1a antigen, MHC Class II antigens, adenosine
triphosphatase) or S-100 protein in their cytoplasm (also found in
melanocytes). Langerhans cells have a key role in antigen presentation.
Dermal dendritic cells
These poorly characterized cells are found around the tiny blood vessels of the papillary dermis. They bear MHC Class II antigens on their surface and, like Langerhans cells, probably function as antigen-presenting cells.
T lymphocytes
These
develop and acquire their antigen receptors (T-cell receptors, TCR) in the
thymus. They differentiate into subpopulations, recognizable by their different
surface molecules (cluster of differentiation markers), which are functionally
distinct.
T-helper (TH)/inducer cells
These
help B cells to produce antibody and also induce cytotoxic T cells to recognize
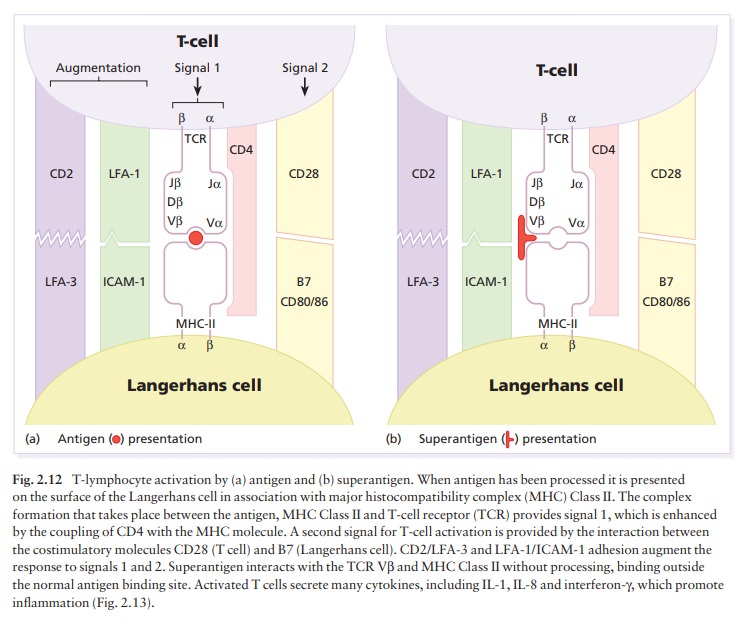
and kill virally infected cells and allogeneic grafts. TH cells recognize
antigen in association with MHC Class II molecules (Fig. 2.12) and, when
triggered by antigen, release cytokines that attract and activate other
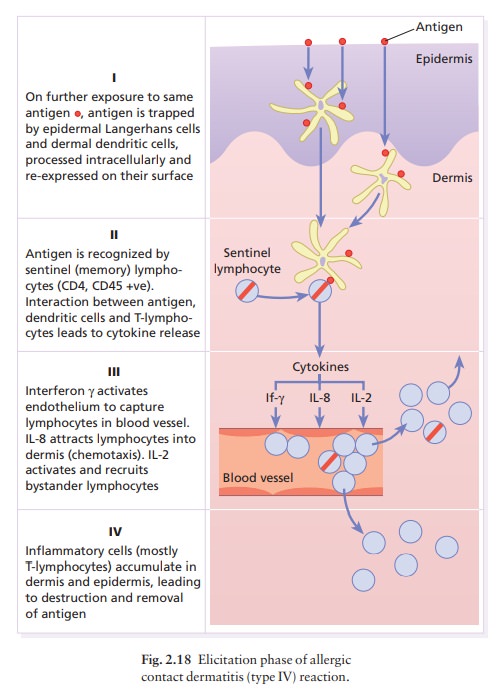
inflammatory cells (see Fig. 2.18). They are CD4+.Helper
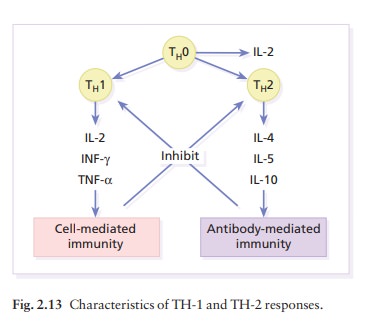
T cells are divided into type 1 (TH-1) and type 2 lymphocytes (TH-2) according
to the main cytokines that they produce (Fig. 2.13). Some skin diseases
dis-play a predominantly TH-1 response (e.g. psoriasis), others a mainly TH-2
response (e.g. atopic dermatitis).
T-cytotoxic (TC) cells
These
lymphocytes are capable of destroying allogeneic and virally infected cells,
which they recognize by the MHC Class I molecules on their surface. They are
CD8+.
T-cell receptor and T-cell gene receptor rearrangements
Most
T-cell receptors are composed of an α
and β chain,
each with a variable (antigen binding) and a constant domain, which are
associated with the CD3 cell surface molecules (Fig. 2.12). Many different
com-binations of separate gene segments, termed V, D and J, code for the
variable domains of the receptor. An analysis of rearrangements of the gene for
the recep-tor is used to determine whether a T-cell infiltrate is likely to be
malignant or reactive. The identification of a specific band, on analysis of
DNA from the lesion, which is not matched by the patient’s DNA from other
sites, indicates monoclonal T-cell proliferation, and suggests either
malignancy or a T-cell response to a single antigen.
L cells/null (non-T, non-B) cells
These
leucocytes have properties between those of T and myelomonocytic cells. Most
have receptors for FcIgG. This subpopulation contains natural killer (NK) and
killer (K) cells.
Natural killer cells
These
are large granular leucocytes that can kill virally infected cells, or tumour
cells that have not previously been sensitized with antibody.
Killer cells
These
are not a separate cell type, but rather cytotoxic T cells, NK cells or
monocytic leucocytes that can kill target cells sensitized with antibody. In
antibody-mediated cellular cytotoxicity, antibody binds to anti-gen on the
surface of the target cell: the K cell binds to the antibody at its other (Fc)
end by its Fc receptor and the target cell is then lysed.
Mast cells
These
are present in most connective tissues, pre-dominantly around blood vessels.
Their numerous granules contain inflammatory mediators (see Fig. 8.1). In
rodentsaand probably in humansathere are two distinct populations of mast
cells, connective tissue and mucosal, which differ in their staining
properties, content of inflammatory mediators and proteolytic enzymes. Skin
mast cells play a central part in the pathogenesis of urticaria.
Related Topics