Chapter: Genetics and Molecular Biology: Attenuation and the trp Operon
Serendipitous Discovery of trp Enzyme Hypersynthesis
The Serendipitous Discovery of trp Enzyme Hypersynthesis
The discovery of polarity is an illustration of the
fact that frequently the most important discoveries result from attempting to
solve some other problem. Polarity is the decrease in the expression of a gene
downstream in an operon from a nonsense mutation. This phenomenon commanded the
attention of many molecular biologists from 1970 to about 1976. Experiments by
Yanofsky and Jackson at this time were designed to study the phenome-non of
polarity by locating within the trp
operon any elements that affected polarity. They sought to isolate deletions
that eliminated the polar effects of a mutation near the beginning of the
operon on the expression of a gene near the end of the operon. As deletions are
rare to begin with, special selections and scorings must be used to identify
cells containing the desired deletion. The basic selection method was to use
conditions in which polarity reduced expression of a promoter-distal trp gene to the extent that cells could
not grow and then to select for secondary mutants that could grow. Among them
would be deletions
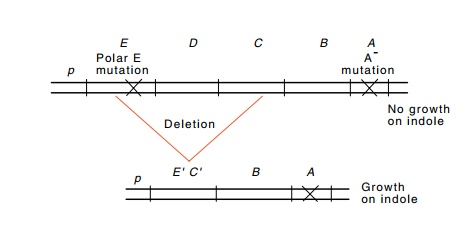
Figure
13.6 How an internaltrpdeletion can relieve polarity
generated by apolar trpE mutation and
increase expression of the trpB gene.
removing any elements that generated polarity.
Scoring by replica plating onto petri plates spread
with lawns of phage carrying parts of the trp
operon then identified the candidates containing deletions internal to the trp operon.
The last
two genes of the tryptophan operon, trpA
and trpB, code for the α and β subunits of tryptophan
synthetase. This enzyme normally has an α2β2
structure. The synthetase from a trpA
mutant has a β2
structure. This dimer possesses part of the enzyme’s normal activity and
catalyzes the conversion of indole and serine to tryptophan and water at about
3% the rate that the wild-type enzyme catalyzes the conversion of serine and
indole-3-glycerol phosphate to tryptophan and water. The activity of the β2 complex
is such that if indole is present in the growth medium, tryptophan can be synthesized
at a rate sufficient to supply the cells’ tryptophan requirements. The
reduction in expression of the trpB gene
in a trpA mutant strain containing a
polar trpE mutation leavesthe cells
unable to convert indole to tryptophan at a rate adequate for growth (Fig.
13.6). Under these conditions, a deletion of the trpE mutation that does not include the trp promoter and the trpB
gene relieves the polarity on trpB
expression and permits cells to satisfy their tryptophan requirements with
indole. Amongst the indole+ colonies will be deletions of various
sizes. One test was determining their sizes.
Candidates
were scored for loss of the trpD and trpC genes with special phage that
carried copies of these genes, but with mutations in the genes. The phage can infect
cells, but many cells are not lysed. Often the phage either recombine into the
chromosome or simply remain in the cell for several generations until diluted
away by cell growth. While present in the cells, the phage can recombine with
homologous DNA on the host chromosome if it is present. Thus, cells that retain
trpD or trpC can reconstruct a functional trpD or trpC genes on
some copies of the phage. These can subsequently be detected by replica plating
onto another lawn of trpC or trpD mutant cells. By these means
Jackson and Yanofsky identified many internal deletions in the trp operon that relieved polarity. We might have expected that fusions of the trpB gene
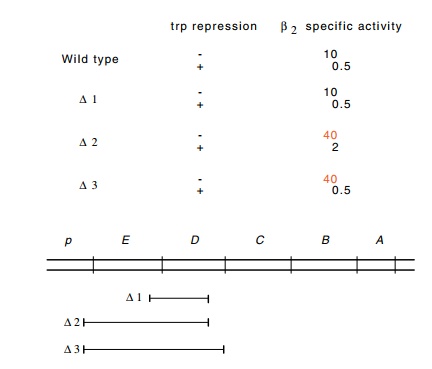
Figure
13.7 The specific activity of theβ2 protein of thetrpoperon
in thepresence and absence of trp
repressor in wild-type cells and in cells containing the three different
deletions indicated.
Out of 34 internal deletions that had removed
polarity on the expres-sion of the trpB
gene, all were still regulated by the Trp repressor. Most unexpectedly, two
deletion strains hypersynthesized the TrpB protein by a factor of three to ten;
each of these was deleted of all of the trpE
gene (Fig. 13.7). The source of this hypersynthesis was then examined more
carefully, as described in the next section.
Related Topics