Matter Around Us | Term 1 Unit 3 | 6th Science - Separation of Mixtures | 6th Science : Term 1 Unit 3 : Matter Around Us
Chapter: 6th Science : Term 1 Unit 3 : Matter Around Us
Separation of Mixtures
Separation of Mixtures
Are all mixtures used as they are? or
is there a need for separating components?
Materials we use in our day-to-day
life are got from different sources and are very often combined with other
substances.
Mixtures like coffee and ice cream are
taken as such. There is no need for separation in these cases. Metals occur in
the form of ores under the earth’s crust. But if we want to use a pure metal,
we need to adopt a laborious process of extraction to separate the useful metal
from the ore.
So what is meant by separation ?
The process by which the components of
mixture are isolated and removed from each other to get pure substance is
called separation.
To get the original properties and
uses of substance we need separation
When and why do we need to separate
mixtures?
* when we need to remove impurities or
harmful components from the mixtures (eg: stones from rice)
* when the useful component has to be separated
from other components (eg: petrol from petroleum)
* when a substance has to be obtained
in highly pure form (eg: gold from gold mines)
Let us visit Selvi’s Family
It is 7 am and
Selvi’s family is busy. At home, in the kitchen, Selvi’s mother is making tea
for the family and her grandmother is separating butter from curds. Her father
and uncle are out in the fields collecting paddy after harvesting. Selvi is
helping her mother in preparing to cook rice and is separating stones from the
rice. Selvi’s little brother Balu is fascinated by a piece of magnet that he
was given by his friend and is playing outside in the sand with it.
Can you list out in
your note book, the different activities that Selvi’s family is engaged in?
Let us explore the
different separating methods involved in the above activities and also learn
about a few other methods.
The choice of
method of separation depends upon the properties of the components of the
mixture. The separation method may be based on the particle’s size, shape or
physical state – they may be solids, liquids or gases.
Selvi’s mother used
a strainer to remove the tea leaves to get the clear liquid. Larger sized
particles of tea leaves will be retained by the strainer while the clear liquid
will pass through. This is called filtering.
Will you discard
the tea leaves after straining? Can you suggest a good way of using them?
A sieve is similar
to a strainer. Sieving is used when we have to separate solid
particles of different sizes. Eg: bran from flour, sand from gravel etc.
Activity
6
Think
and find out!: Is it a good idea to separate bran from flour? Write your answer
in your notebook

Answer: Yes, It is a good idea to separate bran from flour.

Wire
mesh as a strainer sieve is used to separate gravel from sand at a construction
site.
When very fine
insoluble solids have to be separated from a liquid as in butter from curds, Churning
is performed. The mixture is churned vigorously when solid butter will be
collected on the sides of the vessel. Both butter and buttermilk are obtained
after churning are useful and can be consumed.
In washing machines this principle is
used to squeeze out dirt from clothes and the method is called centrifugation.

Threshing
We pluck flowers
from a plant? When we pluck flowers from plants, we are separating the flowers
from their stalks. Can we do the same for food grains like rice and wheat which
also grow on plant stalks? That would not be possible because the grains are
small in size and also the quantity is very large. Farmers separate grains from
their stalks by beating them so hard that the grains are separated from their
stalks. This is called Threshing.

Activity-7
Have
you seen how some farmers spread the dried grain stalks in the middle of the
road? Heavy vehicles pass on them and the grains get separated from the stalks.
Write down two merits and demerits of this practice.
Winnowing
Rice, wheat and
other food grains are covered in husk which cannot be eaten by us. Husk is very
light and gets easily blown away by a breeze or wind. This property is made use
of in winnowing. This is done by dropping the mixture slowly from a
height in the presence of wind. Lighter solids i.e. husks will be carried by
wind and will be collected in a separate heap while heavier solids i.e. grains
will fall closer and form a separate heap.

Rice husks are the hard protecting coverings
of grains of rice. In addition to protecting rice during the growing season,
husks can be used as building material, fertilizer, insulation material and
fuel
If the rice that we
cook has stones in it we have to pick the stones out.
Handpicking
How do we identify
a stone from a grain of rice? If the stones are visibly very different from the
grain, they can be easily picked and separated by hand. This is handpicking.
But if the stones look very similar to the rice grains it is difficult
to separate.

Magnetic
Separation
In a mixture
containing iron, the magnetic property of iron can be used to separate it from
non- magnetic substances by using a magnet. Substances that are attracted to a
magnet are called magnetic. Separating solids using a magnet is called magnetic
separation.

Sedimentation
Rice and pulses are
often mixed with very fine straw, husk or dust particles which have to be
removed before cooking. Are you familiar with the way this is done at home? The
rice or pulses are washed in water. The lighter impurities float while heavier rice
grains sink to the bottom. This is called sedimentation. The water with
the impurities is carefully poured away leaving clean rice at the bottom. This
is called decantation.
SEPARATING
MUD FROM MUDDY WATER: (can be demonstrated by the teacher)
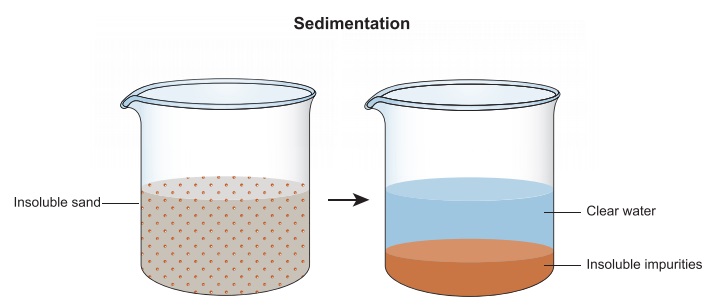
Muddy water is a
mixture of very fine particles of soil in water. What will happen to a beaker
of muddy water if it is left undisturbed for some time? Mud being heavy will
settle down at the bottom of the beaker and will form the sediment. Water will
form the top layer and is called the supernatant liquid.
The settling down
of heavier component of a mixture when allowed to remain undisturbed for some
time is called sedimentation.
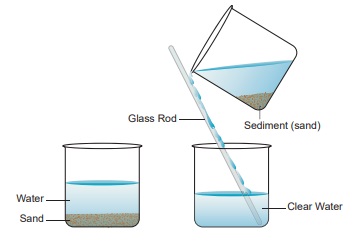
Decantation : This process is done after sedimentation.
The supernatant liquid is slowly poured out from the container without
disturbing the sediment. The part that has settled down is called sediment. The
water that is obtained after decantation is called the decant ate.
The process of
removal of water above the sediment is called decantation. But even after
decantation the water is not completely free from fine soil particles. How can
we remove this? By the process of filtration. Do you think a
strainer or a cloth can filter theses very fine particles? Do it and see for
yourself.
Filtration
We will use filter
papers to remove the finer impurities. A filter paper has very fine pores much
smaller than soil particles. Let us see how to use the filter paper.
Take a piece of
filter paper. Fold it to make a cone (see figure)
Slowly pour the
muddy water over the filter paper. On filtration clear water (filtrate) flows down the funnel and mud settles as residue on
the filter paper.
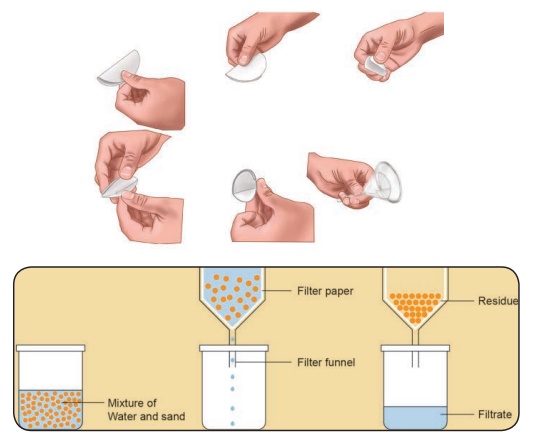
The method of
separating insoluble component (sand, mud etc.) from a mixture using a filter
paper is called filtration.
The liquid which
passes through the filter and comes down is called filtrate and the insoluble
component left behind on the filter is called residue.
More
to know:
Combination
of methods are used sometimes for complete separation.
If
the mixture of sand and salt in water has to be separated several steps will be
involved: Sedimentation, decantation, filtration, evaporation and condensation.
Activity
8
Group
Activity – Students are divided into 4 groups
Each
group should suggest a method to separate mixtures and also give reasons why
they used a particular method and what property of the components forms the
basis for separation. Examples should be drawn from day-to-day life. After
the group presents its method to the rest of the class, the whole class
will discuss and analyse if the suggested method will work and then make a note
of it in the form given below:
Related Topics