Matter Around Us | Term 1 Unit 3 | 6th Science - Diffusion | 6th Science : Term 1 Unit 3 : Matter Around Us
Chapter: 6th Science : Term 1 Unit 3 : Matter Around Us
Diffusion
Diffusion
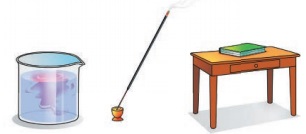
Let us place a book on a table. Let it not be
disturbed. Observe for five minutes.
Now take a glass of water and add just a drop of
ink carefully at the centre. Do not shake or stir.
Now light an incense stick and keep in one corner
of the room.
Let us answer the following questions.
Did the book move?
Did the ink particles move and spread itself in
the water? How long did it take for complete mixing?
Did you get the smell of the incense stick from
where you are standing?
How fast did you get the smell? How did the smell
reach you?
We may conclude
that the particles of gases and liquids can move and that among gases more
easily. We call this movement as diffusion. Diffusion is the
tendency of particles to spread out in order to occupy the available space.
Fact files

Movement of
particles is restricted in solids and they do not diffuse like liquids or
gases. In fluids the particles are under motion and hence can push ink or smoke
particles here and there.
Activity
4
Lift
an uninflated cycle tube. Inflate it and lift it again. Is there a change in
the weight? Can we see that air has mass?
So we
can say that air is also matter though we cannot see.
Let
us try to know more about matter.
Test Yourself
Name an object
which is brittle and transparent.
Name an object
which can be stretched.
Name two objects
which can be bent.

Activity
5
Let
us take two sachets of juice. In both the sachets, it is written 100ml. Let us
empty two sachets and pour the juice into the following glasses.

Does
its shape also change? Yes / No
Answer: Yes
Does
its volume change? when it is poured into a big glass or a small one? Yes / No
Answer: No
How
will you find out whether the volume has changed or not?
Answer: A liquid needs a container and takes the shape of a container because
the particles slide past one another and keep moving. The amount of juice is the
same in both glasses. The volume of a liquid remains the same whether it is kept
in a large container or a small one but its shape changes.
A
liquid needs a container and takes the shape of a container because the
particles slide past one another and keep moving. The amount of juice is the
same in both glasses. The volume of a liquid remains the same whether it is
kept in a large container or a small one but its shape changes.
Try
to draw the shape of the liquid containers alone in your note book. What do you
observe?
Answer: Its
shape changes.
Think
to learn
Solid
→ Liquid → Gas
“Liquefaction
of gases” is the process by which substances in their gaseous state are
converted to the liquid state. When pressure on a gas in increased, its
molecules comes closer together, and its temperature is reduced, which removes
enough energy to make it change from the gaseous to the liquid state.
Related Topics