Chapter: Biochemistry: Transcription of the Genetic Code: The Biosynthesis of RNA
Ribozymes
Ribozymes
There
was a time when proteins were considered the only biological macromolecules
capable of catalysis. The discovery of the catalytic activity of RNA has thus
had a profound impact on the way biochemists think. A few enzymes with RNA
components had been discovered, such as telomerase and RNase P, an enzyme that
cleaves extra nucleotides off the 5' ends of tRNA precursors. It was later
shown that the RNA portion of RNase P has the catalytic activity. The field of
catalytic RNA (ribozymes) was launched in earnest by the discovery of RNA that
catalyzes its own self-splicing. It is easy to see a connection between this
process and the splicing
What are the characteristics of a ribozyme?
Several
groups of ribozymes are known to exist. In Group
I ribozymes, there is a requirement for an external guanosine, which
becomes covalently bonded to the splice site in the course of excision. An
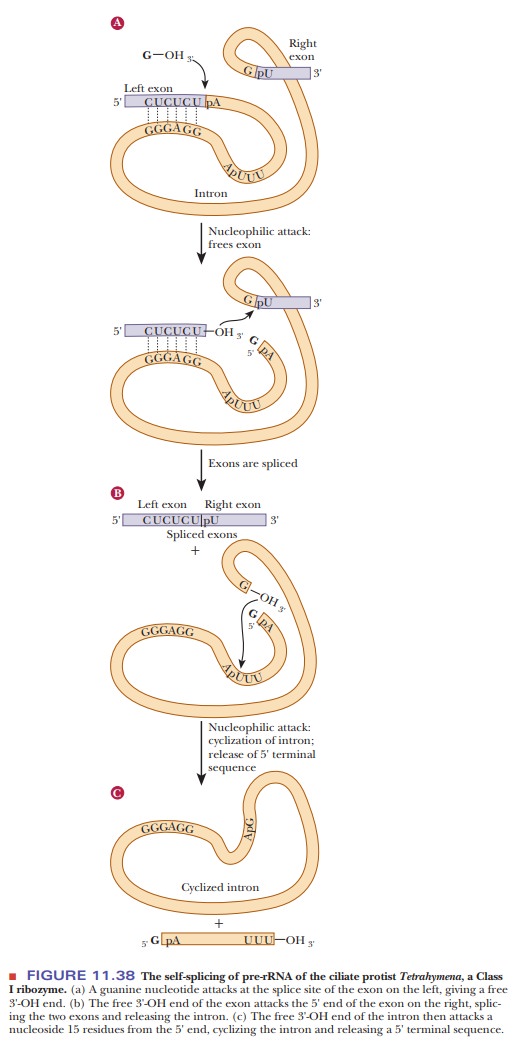
example is the self-splicing that takes place in pre-rRNA of the ciliate
protist Tetrahymena (Figure 11.38).
The transesterification (of phosphoric acid esters) that takes place here
releases one end of the intron. The free 3'-OH end of the exon attacks the 5'
end of the other exon, splicing the two exons and releasing the intron. The
free 3'-OH end of the intron then attacks a nucleotide 15 residues from the 5'
end, cyclizing the intron and releasing a 5' terminal sequence. The precision
of this sequence of reactions depends on the folded conformation of the RNA,
which remains internally hydrogen bonded throughout the process. In vitro, this
catalytic RNA can act many times, being regenerated in the usual way for a true
catalyst. In vivo, however, it appears to act only once by splicing itself out.
Group II ribozymes display a lariat
mechanism of operation that was facilitated by snRNPs. There is no requirement
for an external nucleotide; the 2'-OH of an internal adenosine attacks the
phosphate at the 5' splice site. Clearly, DNA cannot self-splice in this
fashion because it does not have a 2'-OH.
The folding of the RNA is crucial to its catalytic activity, as is the case with protein catalysts. A divalent cation (Mg2+ or Mn2+) is required; it is quite likely that metal ions stabilize the folded structure by neutralizing some of the negative charges on the phosphate groups of the RNA. A divalent cation is essential for the functioning of the smallest ribozymes known, the hammerhead ribozymes, which can be catalytically active with as few as 43 nucleotides. (The name comes from the fact that their structures resemble the head of a hammer when shown in conventional representations of hydrogen-bonded secondary structure.) The folding of RNA is such that large-scale conformational changes can take place with great precision. Similar large-scale changes take place in the ribosome in protein synthesis and in the spliceosome in the processing of mRNA. Note that they remain RNA machines when proteins have taken over much of the catalytic functioning of the cell. The ability of RNA to undergo the requisite large-scale conformational changes may well play a role in the process. A recently proposed clinical application of ribozymes has been suggested. If a ribozyme can be devised that can cleave the RNA genome of HIV, the virus that causes AIDS, it will be a great step forward in the treatment of this disease. Research on this topic is in progress in several laboratories.
Summary
Proteins are not the only biological molecules
with catalytic properties. Some RNAs, called ribozymes, also catalyze certain
reactions.
Group I
ribozymes require an external guanosine for reactivity. Group II ribozymes do
not have this requirement. They carry out catalysis via a lariat mechanism.
Related Topics