Chapter: Essential Clinical Immunology: Immunological Aspects of Cardiac Disease
Rheumatic Fever
RHEUMATIC FEVER
Acute rheumatic fever (ARF) is a delayed, nonsuppurative sequela of a pharyngeal infection with the group A streptococcus. A latent period of two to three weeks fol-lows the initial streptococcal pharyngitis. The latent period remains the same for each individual patient in the event of a recurrence. This suggests that the patient has already been exposed to more than one streptococcal infection in the past. The onset of disease is usually characterized by an acute febrile illness, which may mani-fest itself in one of three classical ways: (1) The patient may present with migratory arthritis predominantly involving the large joints of the body. (2) There may also be
concomitant clinical and laboratory signs of carditis and valvulitis, or carditis and valvulitis may be the only signs of an acute episode. (3) There may be involvement of the central nervous system, manifesting itself as Sydenham’s chorea. The clinical episodes are self-limiting but damage to the valves may be chronic and progressive, resulting in cardiac decompensation and death.
Although both the severity and mor-tality of the disease have dramatically declined since the turn of the century, recent reports indicate a resurgence in this country (Veasy et al. 1987) and in many military installations in the world, remind-ing us that ARF remains a public health problem even in developed countries. In addition, the disease continues essentially unabated in many of the developing coun-tries. Estimates suggest 10–20 million new cases per year in those countries where two-thirds of the world’s population lives.
Epidemiology
The incidence of ARF actually began to decline long before the introduction of antibiotics into clinical practice, decreasing from 250 to 100 patients per 100,000 population from 1862 to 1962 in Denmark (see Figure 12.1). Why the decline occurred is not entirely clear, but better hygiene, improved economic conditions, and less crowding were certainly important factors.
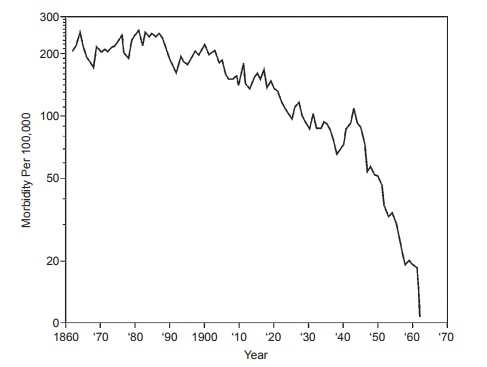
Figure 12.1 Data demonstrating the decline of mortality rates of rheumatic fever long before the advent of penicillin and then the precipitous drop after its introduction.
The introduction of antibiotics in 1950 rap-idly accelerated this decline, until by 1980, the incidence ranged from 0.23 to 1.88 patients per 100,000, primarily in children and teenagers in the United States. A nota-ble exception has been in the Australian aborigines (125/100,000) and the native Hawaiian and Maori populations (both of Polynesian ancestry), where the incidence continues to be 13.4/100,000 hospitalized children per year.
Whether certain M-type strains are more “rheumatogenic” than others remains controversial. Certain investigators have stated that M protein types 5, 14, 18, and 24 are mainly associated with outbreaks, but in the large series collected in the Great Lakes Naval Station of ARF during World War II, the main strains were types 17, 19, and 30. In the Caribbean and in Latin America, the main ARF strains are 11 and 41. Thus, whether certain strains are more “rheumatogenic” than others remains unresolved. What is true, however, is that a streptococcal strain capable of causing a well-documented pharyngitis is generally capable of causing ARF.
Pathogenesis
Although there is little evidence for the direct involvement of group A streptococci in the affected tissues of ARF patients, a large body of epidemiological and immu-nological evidence indirectly implicates the group A streptococcus in the initiation of the disease process: (1) It is well known that outbreaks of ARF closely follow epi-demics of either streptococcal sore throats or scarlet fever. (2) Adequate treatment of a documented streptococcal pharyngitis markedly reduces the incidence of subsequent ARF. (3) Appropriate anti-microbial prophylaxis prevents the recurrences of disease in known patients with acute ARF.
A note of caution is necessary concerning documentation (either clinically or micro-biologically) of an antecedent streptococ-cal infection. The frequency of isolation of group A streptococci from the oropharynx is extremely low during the acute stage of rheumatic fever even in populations with limited access to antibiotics. Further, there appears to be an age-related discrepancy in the clinical documentation of an ante-cedent sore throat. In older children and young adults, the recollection of a strepto-coccal sore throat approaches 70 percent; in younger children, this rate approaches only 20 percent. Thus, it is important to have a high index of suspicion of ARF in children or young adults presenting with signs of arthritis or carditis even in the absence of a clinically and microbiologically docu-mented sore throat.
Another intriguing, yet unexplained observation has been the invariable asso-ciation of ARF only with streptococcal pharyngitis. Although there have been many outbreaks of streptococcal skin infec-tions (impetigo), ARF almost never occurs following infection with these strains. Furthermore, in Trinidad, where both impetigo and ARF are concomitant infec-tions, the strains colonizing the skin are dif-ferent from those associated with ARF and did not influence the incidence of ARF.
Group A Streptococcus
Figure 12.2 is a schematic cross section of the group A streptococci. Many of the anti-gens present in the group A streptococcus cross-react with antigens present in mam-malian tissues: a concept we call molecular mimicry. Thus, antibodies present in the sera of ARF patients bind to cardiac tissue or cells in the caudate nucleus or to human kidney tissues. The controversial question is whether any or some of these antibodies play a role in initiating the disease process. This will be discussed in more detail in the etiology section. For a more detailed dis-cussion of these cross-reactions, the reader is referred to several excellent reviews of this subject matter.
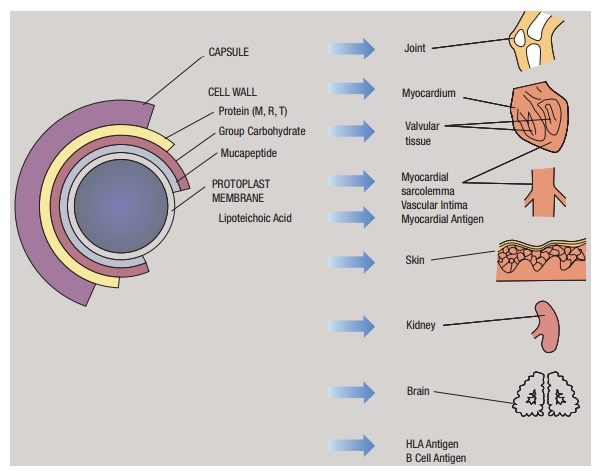
Figure 12.2 Schematic representation of the various structures of the group A streptococcus. Note the wide variety of cross-reactions between its antigens and mammalian tissues.
Although, numerous attempts to pro-duce antibodies to this capsule have been unsuccessful, more recently reports have emerged demonstrating high antibody titers to hyaluronic acid using techniques designed to detect nonprecipitating anti-bodies in the sera of immunized animals. Similar antibodies have been noted in humans. The data establishing the impor-tance of this capsule in human infections have been almost nonexistent, although Stollerman has commented on the pres-ence of a large mucoid capsule as being one of the more important characteristics of certain “rheumatogenic” strains.
Genetics
That ARF might be the result of a host genetic predisposition has intrigued inves-tigators for over a century. It has been variously suggested that the disease gene is transmitted in an autosomal dominant fashion, in an autosomal recessive fashion with limited penetrance, or that it is pos-sibly related to the genes conferring blood-group secretor status.
Renewed interest in the genetics of ARF occurred with the recognition that gene products of the human MHC (major his-tocompatibility complex) were associated with certain clinical disease states. Using an alloserum from a multiparous donor, an increased frequency of a B-cell alloantigen was reported in several genetically distinct and ethnically diverse populations of ARF individuals and was not MHC related.
Most recently, a monoclonal antibody (D8/17) was prepared by immunizing mice with B cells from an ARF patient. A B-cell antigen identified by this antibody was found to be expressed on increased num-bers of B cells in 100 percent of rheumatic fever patients of diverse ethnic origins and only in 10 percent of normal individuals. The antigen defined by this monoclonal antibody showed no association with or linkage to any of the known MHC hap-lotypes, nor did it appear to be related to B-cell activation antigens.
These studies are in contrast to other reports in which an increased frequency of certain human leucocyte antigens (HLAs) was seen in ARF patients. There are marked differences in the increased frequency of the HLAs, depending on the racial fea-tures of the patient group. These seem-ingly conflicting results concerning HLA and RF susceptibility prompt speculation that these reported associations might be of class II genes close to (or in linkage dis-equilibrium with) but not identical to the putatative RF susceptibility gene. Alter-natively, and more likely, susceptibility to ARF is polygenic, and the D8/17 antigen might be associated with only one of the genes (i.e., those of the MHC encoding for the D-related [DR] antigens) conferring susceptibility. Although the full explana-tion remains to be determined, the pres-ence of the D8/17 antigen does appear to identify a population at special risk of contracting ARF.
Related Topics