Chapter: Plant Biochemistry: The Calvin cycle catalyzes photosynthetic CO2 assimilation
Reductive and oxidative pentose phosphate pathways are regulated
Reductive and oxidative pentose phosphate pathways are regulated
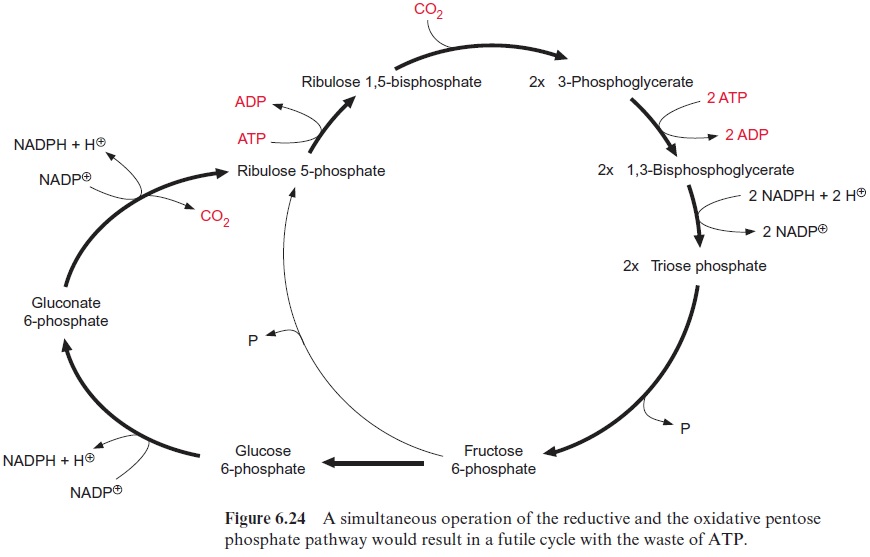
The enzymes of the reductive as well as the oxidative pentose phosphate pathways are located in the chloroplast stroma (Fig. 6.24). A simultane ous operation of both metabolic pathways, in which one molecule of CO2 is reduced to a carbohydrate at the expense of three ATP and two NADPH (reductive pentose phosphate pathway), and then reoxidized by the oxi dative pathway to CO2, yielding two molecules of NADPH, would rep resent a futile cycle. This futile cycle would waste three molecules of ATP in each turn. This is prevented by metabolic regulation, which ensures that key enzymes of the reductive pentose phosphate pathway are active only during illumination and are switched off in darkness, whereas the key enzymes of the oxidative pentose phosphate pathway are only active in the dark.
Reduced thioredoxins transmit the signal “illumination” to the enzymes
An important signal for the state “illumination” is provided by photosyn thetic electron transport as reducing equivalents such as reduced thiore doxin (Fig. 6.25). Electrons of reducing equivalents are transferred from ferredoxin to thioredoxin by the enzyme ferredoxin-thioredoxin reductase, an iron-sulfur protein of the 4Fe-4S type.
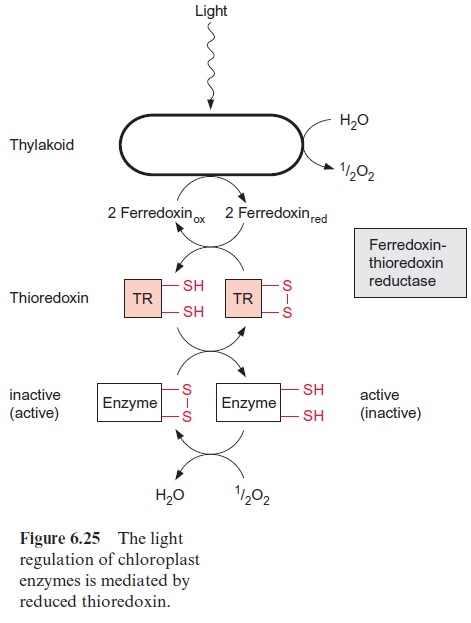
Thioredoxins form a family of small proteins, consisting of about 100 amino acids, which contain as a reactive group the amino acids Cys-Gly-Pro-Cys, located at the periphery of the protein. Due to the neighboring cysteine side chains, the thioredoxin can occur in two redox states: the reduced thioredoxin with two SH-groups and the oxidized thioredoxin in which the two cysteines are linked by a disulfide (S-S) bond.
Thioredoxins are found in all living organisms from archaebacteria to plants and animals. They function as protein disulfide oxido-reductases, in reducing disulfide bonds in target proteins to the -SH form and reoxidizing them again to the S-S form. Despite their small size, they possess a relatively high substrate specificity. Thioredoxins participate as redox carriers in the reduction of high as well as low molecular compounds (e.g., the reduction of ribonucleotides to deoxyribonucleotides; the reduction of sulfate, a process occurring in plants and microorganisms; and the reductive activation of seed proteins during germination). Furthermore, other proc esses are known in which thioredoxins play an essential role, for instance the assembly of bacteriophages, and hormone action or the blood-clotting proc ess in animals.
The involvement of thioredoxins in the light regulation of chloroplast enzymes is a very special function which might have occurred during evo lution in addition to their general metabolic functions. The chloroplast enzymes ribulose phosphate kinase, sedoheptulose 1,7-bisphosphatase, NADP-glyceraldehyde phosphate dehydrogenase and the chloroplast isoform of fruc-tose 1,6-bisphosphatase are converted from an inactive state to an active state via reduction with thioredoxin and are thus switched on by light. This also applies to other chloroplast enzymes such as NADP-malate dehydrogenase and F-ATP synthase . Reduced thioredoxin also convertsRubisCO activase from a less active state to a more active state. Reduced thioredoxin can also inactivate enzymes, e.g., glucose 6-phosphate dehydrogenase, the first enzyme of the oxidative pentose phos phate pathway.
The thioredoxin modulated activation of chloroplast enzymes releases a built-in blockage
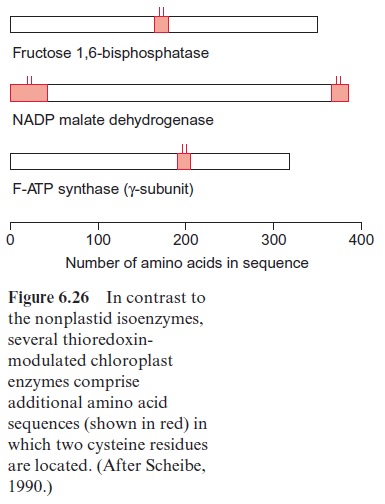
Important knowledge of the mechanism of thioredoxin action on the chloroplast enzymes has been obtained from comparison with the cor responding isoenzymes from other cellular compartments. Isoenzymes of chloroplast fructose 1,6-bisphosphatase, glyceraldehyde phosphate dehydrogenase and malate dehydrogenase exist in the cytosol and are not regulated by thioredoxin. This also applies to F-ATP synthase in the mito chondria. Comparison of the amino acid sequences shows that at least in some cases the chloroplast isoenzymes possess additional sequences at the N- or C-terminus, or in an internal region of their sequence, which provide two cysteine residues(Fig. 6.26). The SH-groups of these cysteine residues can be oxidized and form a disulfide bond, which is the substrate for the disulfide oxidoreductase activity of thioredoxin.
Upon exchange of the cysteine residues involved in the regulation by genetic engineering enzymes were obtained that are fully active in the absence of reduced thioredoxin. Under oxidizing conditions, the enzymes regulated by thioredoxin are forced by the formation of a disulfide bridge into a conformation in which the catalytic center is inac tivated. The reduction of this disulfide bridge by thioredoxin releases this blockage and the enzyme is converted into a relaxed conformation in which the catalytic center is accessible.
The light activation discussed so far is not an all or nothing effect. It is due to a continuous change between the thioredoxin-mediated reduction of the enzyme protein and its simultaneous oxidation by oxygen. The degree of activation of the enzyme depends on the rate of reduction. This is not only due to the degree of the reduction through thioredoxin (and thus to the degree of reduction of ferredoxin), but also to the presence of other metabolites, e.g., the reductive activation of fructose and sedoheptu lose bisphosphatase is enhanced by the corresponding bisphosphates. These effectors cause a decrease of the redox potential of the SH-groups in the cor responding enzymes, which enhances the reduction of the disulfide group by thioredoxin. In this way the activity of these enzymes is increased when the concentration of their substrates rises. Thus the reductive activation of NADP malate dehydrogenase is decreased by the presence of NADP+. This has the effect that the enzyme is only active at a high NADPH/NADP+ ratio. On the other hand, the reductive inactivation of glucose-6-phosphate dehydrogenase is increased by NADPH. Thus with a sufficient supply of NADPH the activity of the oxidative pentose phosphate pathway is turned down. In contrast, the oxidative activation of glucose 6-phosphate dehydro genase is enhanced by NADP+ which increases the activity of the oxidative pentose phosphate pathway when there is a demand for NADPH.
Multiple regulatory processes tune the reactions of the reductive pentose phosphate pathway
An additional light regulation of the Calvin cycle is based on the effect of light-dependent changes of chloroplast enzyme activities due to the stro mal proton and Mg++concentrations. When isolated chloroplasts are illuminated, the acidification of the thylakoid space is accom panied by an alkalization and an increase in the Mg++concentration in the stroma. During the dark/light transition, the pH in the stroma may change from about pH 7.2 to 8.0. This correlates with the pH optimum of the CO2fixation of isolated chloroplasts of about pH 8.0 with a sharp decline towards the acidic range. An almost identical pH dependence is found with the light-activated enzymes fructose 1,6-bisphosphatase and sedoheptulose 1,7-bisphosphatase. Moreover, the catalytic activity of both these enzymes is increased by the light-dependent increase of the stromal Mg++ concen tration. The light activation of these enzymes due to the thioredoxin system and light-induced changes of the stromal pH and Mg++concentration is a very efficient system for switching these enzymes on and off, according to demand. During darkness this system results in an extensive inactivation of the corresponding enzymes.
The activities of several stromal enzymes are also regulated by meta bolite levels. The chloroplast fructose 1,6-bisphosphatase and sedoheptulose 1,7-bisphosphataseare inhibited by their corresponding products, fructose 6-phosphate and sedoheptulose 7-phosphate, respectively. Thus the accumu lation of the products has negative effects on the activity of these enzymes. Ribulose phosphate kinase is inhibited by 3-phosphoglycerate and also by ADP. Inhibition by ADP is important for coordinating the two kinase reac tions of the reductive pentose phosphate pathway. Whereas ribulose phos phate kinase catalyzes an irreversible reaction, the phosphoglycerate kinase reaction is reversible. If both reactions were to compete for ATP in an unre stricted manner, in the case of a shortage of ATP the irreversible phospho rylation of ribulose 5-phosphate would be at an advantage, resulting in an imbalance of the Calvin cycle. A decrease in the activity of ribulose phos phate kinase at an elevated level of ADP can prevent this.
Finally, fructose 1,6-bisphosphatase and sedoheptulose 1,7-bisphos phatase are strongly inhibited by glycerate. Glycerate is an intermediate in the recycling of phosphoglycolate formed by the oxygenase activity of RubisCO. The accumulation of glycerate slows down the regeneration of RuBP and its carboxylation. In this way also the accompanying oxygenation is lowered, decreasing the synthesis of glycolate which is the precursor of glycerate.
Also, RubisCO is subject to regulation. Experiments with whole leaves demonstrated that the degree of the activation of RubisCO correlates with the intensity of illumination and the rate of photosynthesis. The activation state of RubisCO is adjusted via a regulation of the RubisCO activase (sec tion 6.2). RubisCO-activase is activated by reduced thioredoxin and is also dependent on the ATP/ADP ratio. When there is a rise in the ATP/ADP ratio in the stroma, the activity of the activase also rises. This scenario explains how the activity of RubisCO can be adjusted to the supply of ATP delivered by the light reaction of photosynthesis. However, many obser vations suggest that this cannot be the only mechanism for a light regu lation of RubisCO, for example the RubisCO activase is regulated by the light-dependent proton gradient across the thylakoid membrane, and the activity of RubisCO is inhibited by its product 3-phosphoglycerate. In this way the activity of RubisCO could be adjusted according to the product accumulation
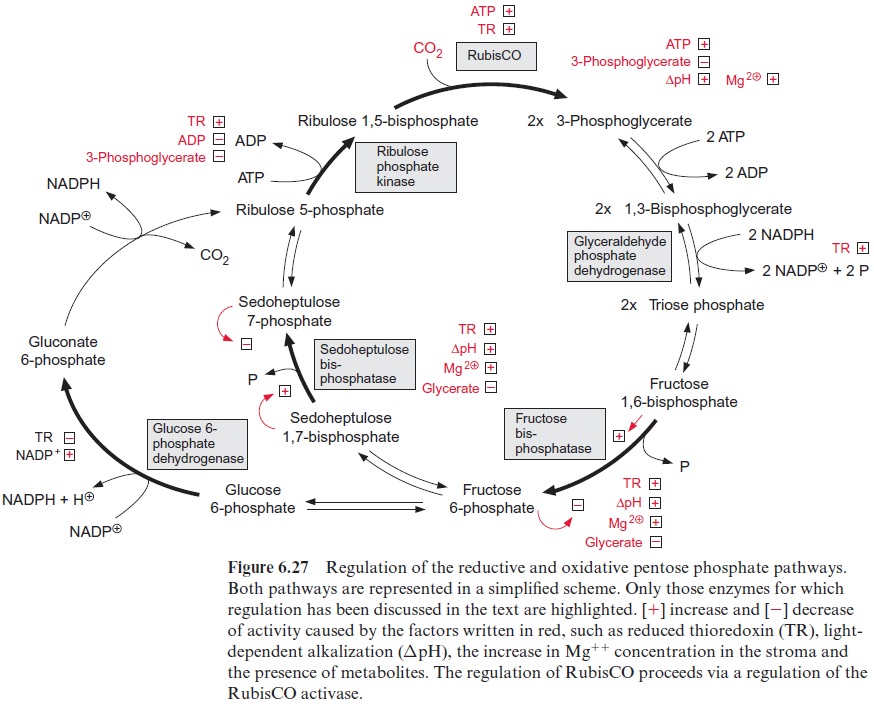
Figure 6.27 presents a scheme summarizing the various factors that influence the regulation of the enzymes of the reductive and oxidative pen tose pathways. A multitude of regulatory processes ensures that the various steps of both reaction chains are adjusted to each other and to the demand of the cell.
Related Topics