Chapter: Physics : Properties of Mater and Thermal Physics
Radial flow of heat
Radial
low of heat
In this method, heat flows from the
inner side towards the other side along the radius of the cylindrical shell.
This method is interesting because
there is no loss of heat as in the other methods.
Cylindrical
Shell method
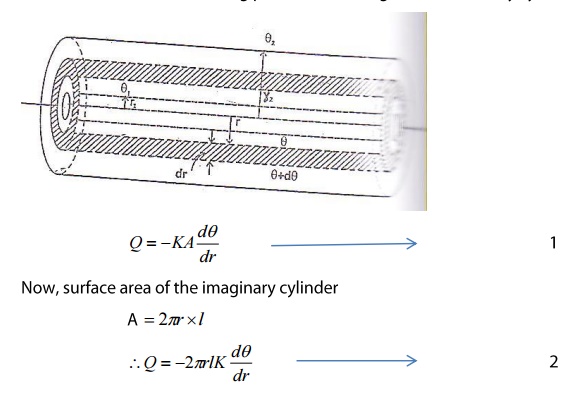
Consider a cylindrical tube of length
l, inner radius r1 and outer radius r2 as shown in the figure. The tube carries
steam or some hot liquid.
Heat is conducted radially across
the wall of the tube. After the steady state is reached, the temperature of the
inner surface θ1 and on the outer surface θ2. This thick
pipe is imagined to consist of a large number of thin coaxial cylinders of increasing
radius. Any such thin imaginary cylinder of the material of thickness ‘dr’ at a
distance r from the axis of the pipe is taken.
Amount of heat flowing per second through this elementary cylinder
After steady state is reached, the
amount of heat lowing(Q) through all imaginary cylinders is same.
Rearranging, the equation 2, we get
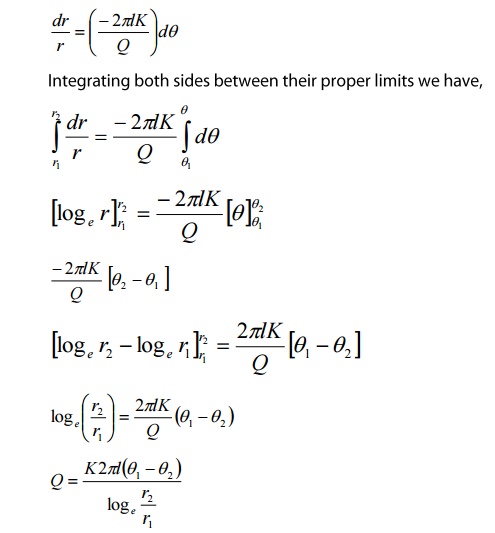
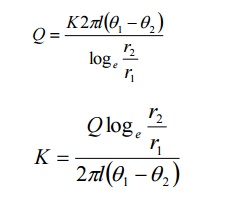
THERMAL
CONDUCTIVITY OF RUBBER
Principle:
It is based on the principle of
radial low of heat through a cylindrical shell.
Procedure:
A big empty calorimeter with
stirrer is weighted (W1). It is then illed with two thirds of water and again weighted(W2).
A known length(l) of a rubber tube is immersed in water in the calorimeter as
shown in the figure.
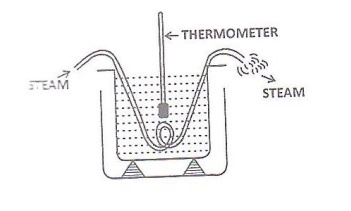
The calorimeter is stirred dwell
and the initial temperature θ1oC is noted. Now one end of
the rubber tube is connected to a steam generator and steam is passed through
it. The steam is passed continuously till there is rise of 10 oC in
temperature. The time taken (t second) or this rise in temperature is noted. The
inal temperature of the water θ2 oC in the calorimeter is
also noted .
Observation
Mass of the empty calorimeter with
stirrer = W1 kg
Mass of the calorimeter with water
= w2 kg
Mass of the water = (W2-W1)
kg
Initial temperature of the water = θ1
oC
Final temperature of the water = θ2
oC
Rise in temperature of the water =
(θ1 -θ2)
Time for which steam is passed = t
seconds
Length o the rubber tube immersed
in water = l
Inner radius of the rubber tube = r1
Outer radius of the rubber tube = r2
Specific heat capacity of Calorimeter = S1
Specific heat capacity of Water = S2
Heat gained by the calorimeter =
Mass x Specific heat capacity x change in temperature
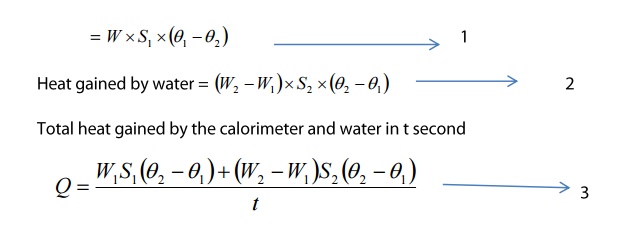
The expression for the thermal
conductivity (K) in the case o cylindrical shell method is given by

Related Topics