Chapter: Physics : Properties of Mater and Thermal Physics
Modes of Heat Transer
MODES
OF HEAT TRANSFER
Heat is one of the forms of energy.
It is transmitted from one place to another by three different ways.
They are
Conduction
Convection
Radiation
1
Thermal conduction
It is well known fact that is
conducted through the material of the body. In conduction, heat transfer takes
place from one point to another through a material medium without the actual
movement of the particles in that medium.
The heat is transmitted from a body
of higher temperature to that o lower temperature.
As an example, when a metal rod is
heated at one end, the heat gradually lows along the length o the rod and the
other end o the rod also becomes hot after some time. This shows that heat has
travelled through the molecules of the rod from one end to other. The molecules
in the rod remain fixed in their mean positions.
On heating the energy molecules
increases and they start vibrating about their mean positions. They collide
with the neighbouring molecules. Because of this collision, the neighbouring
molecules are set into vibration.
Each molecule thus transfers some
of the heat it receives from its predecessor to its successor. Thus the
transmission of heat takes place by molecular vibration in case of conduction.
DEFINITION
It is the process of transmission
of heat from one point to another through substance (or some medium) without
the actual motion o the particles.
Conduction always requires some
material medium. The material medium must be solid. As it requires medium, the
conduction process takes place over vacuum. In fluids (liquid and gas), heat
transmission is through the process of convection.
2
Thermal Conductivity
The ability of a substance to
conduct heat energy is measured by thermal conductivity
EXPRESSION
OR THERMAL CONDUCTIVITY
Consider a slab of material of
length x meter and area of cross section A as shown in the figure.
One end of the slab is maintained
at a higher temperature θ1 and the other end at a lower temperature
is θ1. Heat flows from the hot end to the cold end. It is found that
the amount of heat(Q) conducted from one end to another end is
Directly proportional to the area
of cross-section(A)
Directly proportional to the
temperature dierence between the end θ1- θ2
Directly
proportional to the time of conduction(t)
Directly proportional to the length
of (x).

Where K is the proportionality
constant. It is known as coefficient of thermal conductivity or simple or
thermal conductivity. Its value depends upon the natue of the material.
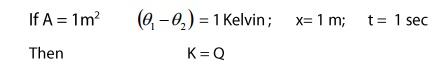
The condition define the
coefficient of Thermal conductivity
Definition:
It is defined as the amount of heat
conducted per second normally across unit area of cross-section of the material
per unit temperature difference per uint length.
The quantity (θ1- θ2)
/ x denote the rate of fall of temperature with respect to distance. It is know
as temperature gradient.

The negative sign indicates the
fall of temperature with distance.
Unit:
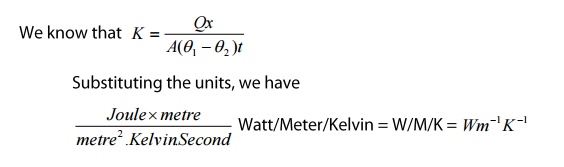
3 Newton's Law of Cooling
Statement:
It state that the rate at which a
body loses heat is directly proportional to the temperature difference between
the body and that of the surrounding.
The amount of heat radiated depends
upon the area and nature of the radiating surface.
If ‘θ’ is the temperature of the
body at any instant and ‘θo’ the temperature of the surroundings, then
according to Newton’s law of cooling, heat lost is proportional to the
difference of temperature between the body and surroundings i.e.( θ1-
θo)
If dQ is the quantity of heat lost
in a small time dt, then
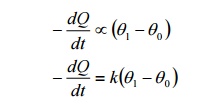
Where k is the constant depending
upon the area and the nature o the radiating surface. The negative sign indicates
that there is decrease of heat with time.
Expression
when a heat body cools from θ1oC to θ2oC
in time t
Consider a body of mass m, specific
heat capacity S and at temperature θ. Suppose the temperature falls by a small
amount dθ in time dt.
Thus, the amount of heat lost

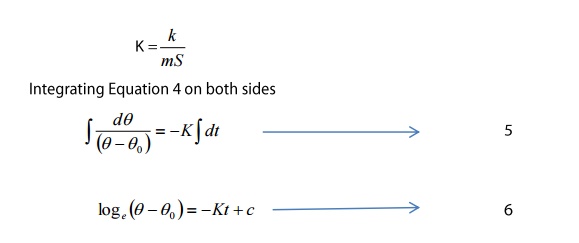
Where c is the constant of integration this
equation is of the form
y=mx+c and it represents a Straight
line.
If the cooling takes place rom θ1o
C to θ2oC in time t then taking the limits, we have

4.
VERIICTION O NEWTON’S LAW O COOLING
The given empty spherical
calorimeter is filled with boiling water and a thermometer is kept in the
orifice as shown in the figure. When the temperature reaches 800C, a stop clock
is started. The time taken for every 2oC fall in temperature is
noted, till the temperature reaches 60 oC.
The rate of cooling at various
temperature is determined. A graph is drawn with rate of cooling along y-axis
and the excess of temperature of the calorimeter over the surrounding along the
x-axis.
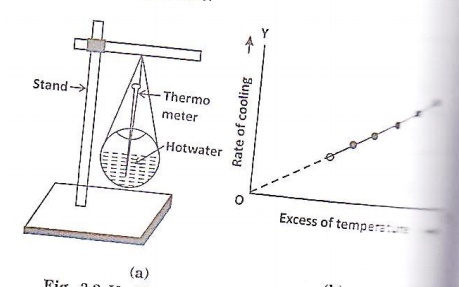
The graph is found to be a straight
line, thereby, showing that the rate of cooling is proportional to the excess
of temperature.
Limitations
The temperature difference between
the hot body and surrounding should be low.
The heat loss is only by radiation
and convection.
The temperature of hot body should
be Uniform throughout.
Applications
The specific heat capacity of the
liquid is determined by using this law.
5
RECTILINEAR FLOW OF HEAT THROUGH A ROD
Consider a long rod AB of Uniform
cross section heated at one end A as shown in the figure. Then there is low of
heat along the length o the bar and heat is also radiated from its surface. B
is the cold end.
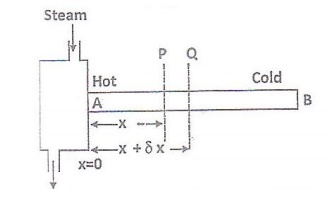
Consider the low of heat between
the section P and Q at distance x and x+δx rom the hot end. Excss temperature
at Section P above the surroundings=θ


6
BEORE THE STEADY STATE REACHED
Before the steady state is reached,
the amount of heat Q is used in two ways. A part of the heat is used in raising
the temperature of the rod and the remaining heat is lost by radiation from the
surface o the element.
Heat absorbed per second to raise
the temperature of the rod


The equation 8 is the standard differential equation for the flow of heat through the rod.
SPECIAL CASE
The thermal conductivity of any
material is determined using equation 8 by considering the actual condition of
the material.
Case
1: When heat is lost by radiation is negligible
If the rod is completely covered by
some insulating materials, then there is no loss of heat due to radiation.
Hence, the heat lost by radiation Epδxθ is zero.
In that case, the total heat gained
by the rod is completely used to raise the temperature of the rod.
From equation 8
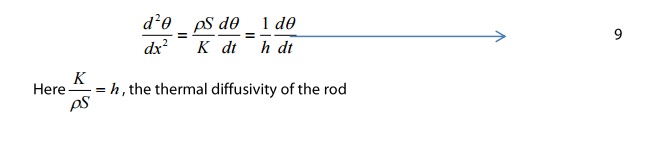
Case
2: After the steady state is reached
After the steady state is reached,
there is no raise of temperature.

Here A and B are two unknown
constants which can be determined from the boundary conditions of the problem.
Suppose
the bar is of infinite length
Excess o temperature above the
surroundings of the hot end = θo
Temperature of the other end (cold
end) = 0
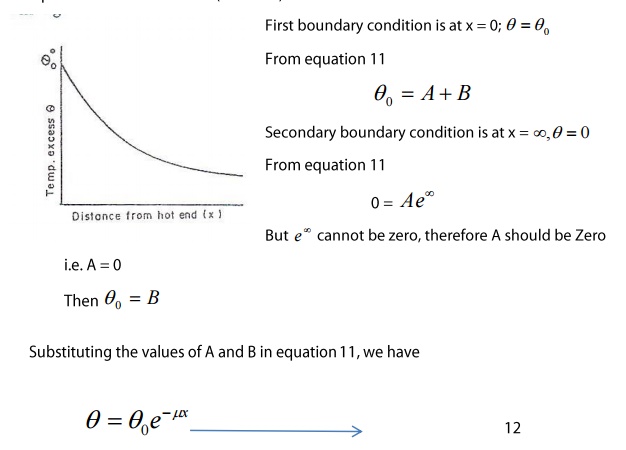
This equation 12 represents the
excess of temperature of a point at distance x from the hot end after the steady
state is reached and it represents an exponential curve.
The temperature falls exponentially
from the hot end as shown in the figure.
7
HEAT CONDUCTION THROUGH A COMPOUND MEDIA OF TWO LAYERS BODIES IN SERIES
Let us consider a composite slab
(or compound wall) of two different materials A and B with thermal
conductivities K1 and K2 and of thickness x1
and x2.
The temperature of the outer aces of A and B are θ1 and θ2.
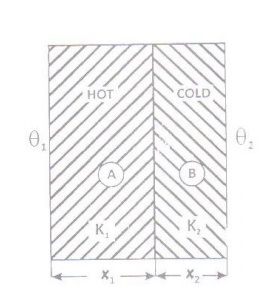
The temperature of the surface in
contact is θ. When the steady state is reached, the amount of heat flowing per
second(Q) through every is same.
Amount of heat flowing through the
material(A) per second

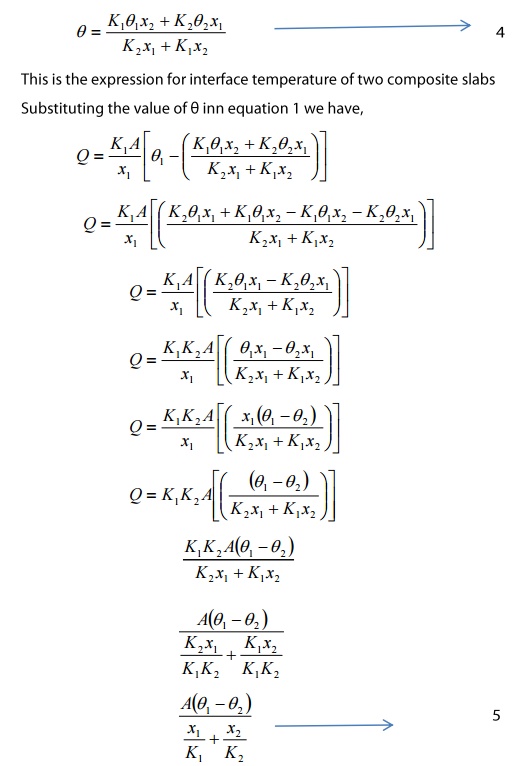
‘Q’ is the value o heat lowing
through the compound wall of the two materials
The method can be extended to
composite slab with more than two slabs.
In general for any number of walls
or slabs, the amount of heat conducted is
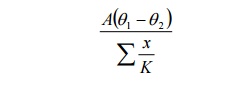
Bodies
in Parallel
Let us consider a composite slab
(or Compound wall) of two dierent materials A and B with thermal conductivities
K1 and K2 and o thickness x1 and x2.
They are arranged in parallel as shown in the figure.
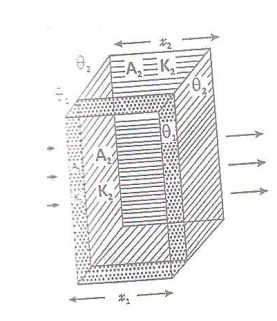
Let the faces of the material be at
temperature θ1 and the respective other end aces be at θ2
temperature.
A1 and A2 be the ares o cross
section of the materials
Amount of heat flowing through the
irst material (A) in one second

8
Methods to determine Thermal conductivity
The thermal conductivity of a
material is determined by different methods.
1. Searle’s
Method – or good conductor like Metallic rod
2. Forbe’s
Method – for determining the absolute conductivity of metal
3. Lee’s
Disc method – for bad conductors
4. Radial
Flow method – or Bad conductors.
Related Topics