Chapter: Biotechnology Applying the Genetic Revolution: Transgenic Plants and Plant Biotechnology
Plant Tissue Culture
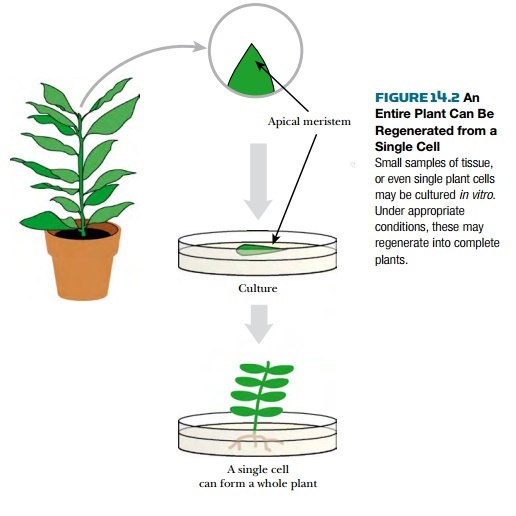
PLANT
TISSUE CULTURE
One major advantage of plants
is that they can often be regenerated from just a single cell, that is, each
plant cell is totipotent and retains
the ability to develop into any cell type of a mature plant (Fig. 14.2). There
is no absolute separation of the germline from the somatic cells in plants
(unlike animals). This unique feature of plants allows scientists to grow and
manipulate plant cells in culture, then regenerate an entire plant from the
cultured cells.
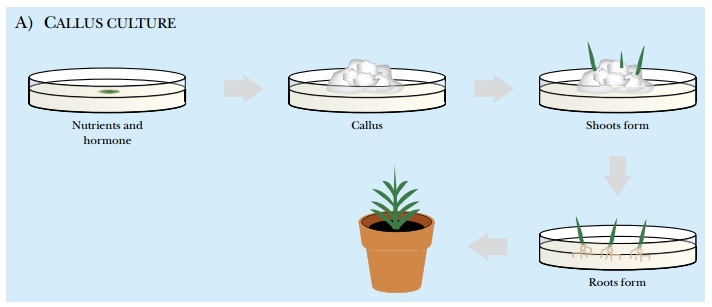
Plant tissue culture can be
done on either a solid medium in a petri dish, called callus culture, or in liquid, called suspension culture. In both cases, a mass of tissue or cells, known as an explant, must be removed from the plant of interest. In callus
culture, the tissue can be an immature embryo, a piece of the apical meristem
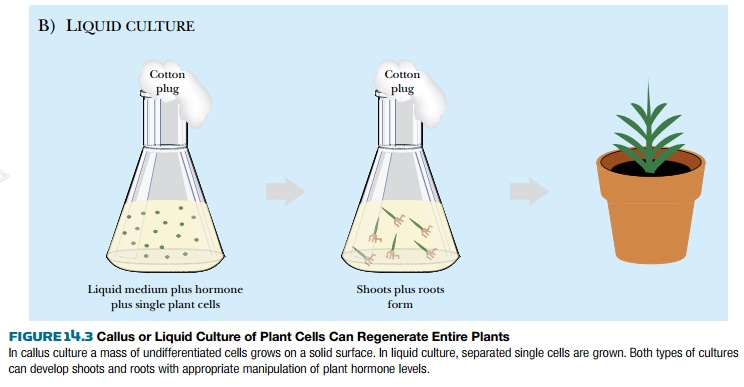
(the region where new plant shoots develop), or a root tip. For liquid culture,
cells must be dissociated from one another. Liquid culture usually uses protoplasts (plant cells from which the
cell wall is removed), microspores (immature pollen cells), or macrospores
(immature egg cells). The cells are then cultured with a mixture of nutrients
and specific plant hormones that induce the undifferentiated cells to grow.
Different types of plants respond to different hormones. To culture wheat cells, for example, the explant is grown with 2,4-dichlorophenoxyacetic acid (2,4-D). This is an analog of the plant hormone auxin, which stimulates plant cells to dedifferentiate and grow. To culture tomato plants, the hormone cytokinin dedifferentiates the cells and induces cell division. In callus culture, undifferentiated cells form a crystalline white layer on top of the solid medium, called the callus. After about a month of growth, the mass of undifferentiated cells can be transferred to medium with a lower concentration of hormone or with a different hormone. Decreasing the amount of hormone allows some of the undifferentiated callus cells to develop into a plant shoot. In most cases the small shoots look like new blades of grass growing from the mass of cells. After another 30 days, the hormone is removed completely, which allows root hairs to start growing from some of the shoots. After another 30 days, small plants can be isolated and planted into soil. In liquid culture, hormones are also used to stimulate the growth of undifferentiated cells, but the shoot and root tissues grow simultaneously (Fig. 14.3).
Because plant tissue culture allows many plants to be produced from one source, the technique is useful for making clones of one particular plant. If a very rare plant is identified, it can be propagated using tissue culture. Only a small cutting is needed to generate many identical progeny. Certain special plant varieties that are hard to maintain by producing seed can be maintained for the long term in culture. Plant cell culture has also been used for mutation breeding. Rather than using seeds, undifferentiated cells are exposed to the mutagen, and plants are regenerated from the mutagenized cells. Mutagens are more effective on the exposed cells of a callus rather than the protected cells within a seed.
Merely growing plants by
tissue culture may induce mutations. The process of regenerating a plant from a
single cell may cause three different types of alterations. Temporary
physiological changes can occur in the regenerated plant. For example, when
blueberry plants are regenerated via tissue culture, the plants are much
shorter. These changes are not permanent, and after a few years growing in the
field, the regenerated blueberries are no different from any other blueberries.
Another alteration that may occur is an epigenetic
change. This is an alteration that
persists throughout the lifetime of the regenerated plant, but is not passed on to the next generation. Epigenetic changes
are often due to alterations in DNA methylation. Finally, true genetic changes affect the regenerated
plant and all its progeny. These may be due to changes in ploidy level,
chromosome rearrangements, point mutations, activation of transposable
elements, or changes in chloroplast or mitochondrial genomes. These types of
changes are relatively common, but changes in the available nutrients and
hormones during tissue culture can dramatically decrease the frequency of
mutation.
Related Topics