Causes and effects of ozone layer depletion - Environmental Issues - Ozone Depletion | 12th Zoology : Chapter 13 : Environmental Issues
Chapter: 12th Zoology : Chapter 13 : Environmental Issues
Ozone Depletion
Ozone Depletion
At about 15 and 30 kilometers from the ground
level, the earth’s atmosphere has a thin layer of ozone, which absorbs
ultraviolet sunlight. Ozone is found in the layer of the atmosphere called the
Stratosphere. It acts as a protective covering that absorbs ultraviolet (UV)
radiation from the sun. The ozone molecule (O3) consists of three
oxygen atoms. It is formed when atmospheric oxygen (O2) on exposure to solar
radiation breaks into two oxygen atoms; each atom then joins up with a single
oxygen atom. The ozone molecule is unstable. It soon decays again to form
molecular oxygen. This cycle is a continuous process in the upper reaches of
the stratosphere.
World Ozone Day
September 16 has been designated by the United
Nations as the International Day for the Preservation of the Ozone Layer.
The ozone layer was discovered in 1913 by the
French physicists Charles Fabry and Henri Buisson.
Causes and effects of ozone layer depletion
Causes: Ozone layer depletion mainly occurs by
anthropogenic actions.
The excessive release of chlorine and bromine
from man-made compounds such as chlorofluorocarbons (CFCs) causes ozone layer
depletion. CFCs, methyl chloroform, carbon tetrachloride,
hydrochlorofluorocarbons, hydrobromofluorocarbons and methylbromide are found
to have direct impact on the depletion of the ozone layer. These are
categorized as ozone-depleting substances (ODS).
Effects: UV rays may penetrate deep into the skin
and can lead to premature skin aging and wrinkling of skin; suppression of the
immune system, skin cancer (melanoma) and chronic effects leading to eye
damage. DNA damage can result from free radicals and reactive oxygen and
photons can damage the DNA itself.
Control: Ozone layer depletion can be controlled
by
1. Phase down or ban the use of CFCs (CFC free
refrigerants).
2. Minimizing the use of chemicals such as
halons and halocarbons.
3. Creating awareness about ozone depleting
agents.
Ozone hole (in purple colour), is the area
above Antarctica, where the ozone layer is the thinnest.
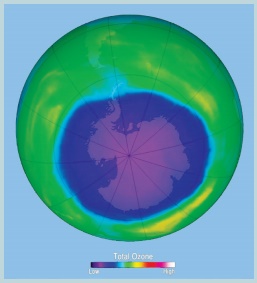
Ozone depletion around Polar region
Ozone thickness is given in Dobson unit (see
carefully the scale shown in colour from violet to red). The ozone hole over
Antarctica develops each year between late August and early October.
Courtesy: NASA
Related Topics