Chapter: Medical Surgical Nursing: Gastrointestinal Intubation and Special Nutritional Modalities
Nursing Management of Patients Undergoing Nasogastric or Nasoenteric Intubation
NURSING
MANAGEMENT OF PATIENTS UNDERGOING NASOGASTRIC OR NASOENTERIC INTUBATION
Nursing
interventions include the following:
•
Instructing the patient about the purpose of the
tube and the procedure required for inserting and advancing it
•
Describing the sensations to be expected during
tube insertion
•
Inserting the NG tube and assisting with insertion
of the nasoenteric tube
•
Confirming the placement of the NG tube
•
Advancing the nasoenteric tube
•
Monitoring the patient and maintaining tube
function
•
Providing oral and nasal hygiene and care
•
Monitoring for potential complications
•
Removing the tube
Providing Instruction
Before
the patient is intubated, the nurse explains the purpose of the tube; this
information may assist the patient to be coopera-tive and tolerant of what is
often an unpleasant procedure. The general activities related to inserting the
tube are then reviewed, including the fact that the patient may have to breathe
through the mouth and that the procedure may cause gagging until the tube has
passed the area of the gag reflex.
Inserting the Tube
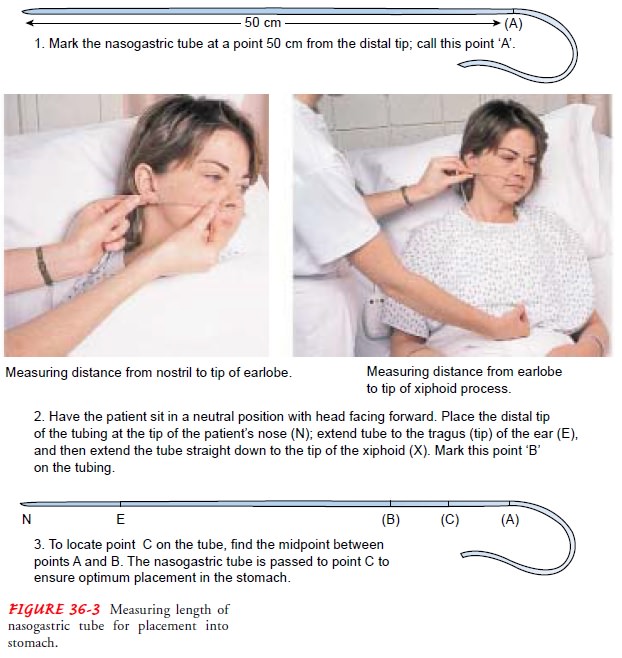
Before
inserting the tube, the clinician determines how much tubing will be needed to
reach the stomach or the small intestine. A mark is made on the tube to
indicate the desired length. This length is determined by measuring the
distance from the tip of the nose to the earlobe, and from the earlobe to the
xiphoid process, then adding 6 inches for NG placement or 8 to 10 inches for
intestinal placement (Fig. 36-3).
While the tube is being inserted, the patient usually sits up-right with a towel spread bib-fashion over the chest. Tissue wipes are made available. Privacy and adequate light are provided. The physician may swab the nostril and spray the oropharynx with Cetacaine (tetracaine/benzocaine) to numb the nasal passage and suppress the gag reflex.
This makes the entire procedure more tol-erable. Having the patient gargle with
a liquid anesthetic or hold ice chips in the mouth for a few minutes can have
the same effect. Encouraging the patient to breathe through the mouth or to
pant often helps, as does swallowing water, if permitted.
A
polyurethane tube may need to be warmed to make it more pliable. To make the
tube easier to insert, it should be lubricated with a water-soluble substance
(K-Y jelly) unless it has a dry coat-ing called hydromer, which, when
moistened, provides its own lubrication. The nurse wears gloves during the
procedure.
The
patient is placed in Fowler’s position, and the nostrils are inspected for any
obstruction. The more patent nostril is selected for use. The tip of the
patient’s nose is tilted, and the tube is aligned to enter the nostril. When
the tube reaches the nasopharynx, the patient is instructed to lower the head
slightly and to begin to swallow as the tube is advanced. The patient may also
sip water through a straw to facilitate advancement of the tube. The oropharynx
is inspected to ensure that the tube has not coiled in the pharynx or mouth.
Confirming Placement
To
ensure patient safety, it is essential to confirm that the tube has been placed
correctly, particularly because tubes may be acci-dentally inserted in the
lungs, which may be undetected in high-risk patients. Examples of high-risk
patients are those with a decreased level of consciousness, confused mental
state, poor or absent cough and gag reflexes, or agitation during insertion.
Pres-ence of an endotracheal tube and recent removal of an endotra-cheal tube
also increase the risk for inadvertent placement of the tube in the lung (Metheny,
1998). Initially, an x-ray study should confirm tube placement. However, each
time liquids or medica-tions are administered, and once a shift for continuous
feedings, the tube must be checked to ensure that it remains properly placed.
The traditional recommendation has been to inject air through the tube while
auscultating the epigastric area with a stethoscope to detect air
insufflations. However, studies indicate that this auscultatory method is not
accurate in determining whether the tube has been inserted into the stomach,
intestines, or respiratory tract (Metheny et al., 1999). Instead of the
auscul-tation method, a combination of three methods is recommended:
•
Measurement of tube length
•
Visual assessment of aspirate
•
pH measurement of aspirate
After
the tube is inserted, the exposed portion of the tube is measured and the
length is documented. The nurse measures the exposed tube length every shift
and compares it with the original measurement. An increase in the length of
exposed tube may in-dicate dislodgement, or a leaking or ruptured balloon if
the tube has a balloon.
Visual
assessment of the color of the aspirate may help iden-tify tube placement.
Metheny et al. (1994) found that gastric as-pirate is most frequently cloudy
and green, tan or off-white, or bloody or brown. Intestinal aspirate is
primarily clear and yellow to bile-colored. Pleural fluid is usually pale
yellow and serous, and tracheobronchial secretions are usually tan or off-white
mucus. Researchers suggest that the appearance of the aspirate may be helpful
in distinguishing between gastric and intestinal placement but is of little
value in ruling out respiratory placement. This method is less helpful when the
patient is receiving continuous feedings, because aspirate often looks like the
formula that is used for the feeding (Metheny & Titler, 2001).
Determining
the pH of the tube aspirate is a more accurate method of confirming tube
placement. The pH method can also be used to monitor the advancement of the
tube into the small intestines. The pH of gastric aspirate is acidic (1 to 5).
The pH of intestinal aspirate is approximately 6 or greater, and the pH of
respiratory aspirate is more alkaline (7 or greater). pH testing is best suited
for distinguishing between gastric and intestinal placement. A pH sensor
enteral tube is available which does not require fluid aspirate to obtain pH
values; it can be useful in dis-tinguishing gastric from small bowel placement
of the tube. The pH method is less helpful with continuous feedings, because
tube feedings have a pH value of 6.6 and neutralize the GI pH (Metheny &
Titler, 2001). For more information, see Nursing Research Profile 36-1.
Using
gastric aspiration as a means of verifying that the NG tube has been placed
correctly may be a problem because of the characteristic properties and
diameter of the tubes. Studies suggest that aspiration may be performed more
easily with polyurethane tubes and tubes with a size 10 Fr diameter. Metheny et
al. (1993) recommended the following steps if problems occur with aspira-tion
of fluid from small-bore feeding tubes:
i) Insufflate 20 mL of air
through the tube with a large syringe (30 to 60 mL).
ii) Pull back on the
plunger.
iii) If step 2 is
ineffective, insufflate another 20 mL of air and re-place the large syringe
with a smaller one (12 mL); attempt to aspirate.
iv) If the measure is still
ineffective, repeat step 3.
v) Change the patient’s
position and attempt to aspirate.
Securing the Tube
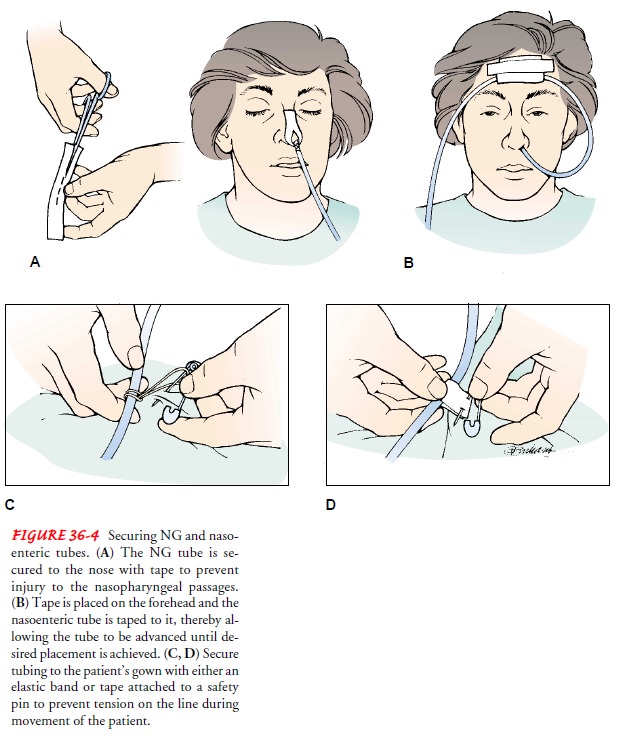
After
the correct position of the tip has been confirmed, the NG tube is secured to
the nose (Fig. 36-4). A liquid skin barrier should be applied to the skin where
the NG tube will be secured. The prepared area is covered with a strip of
hypoallergenic tape or Op-site; the tube is then placed over the tape and
secured with a second piece of tape. The nasoenteric tube can be secured by
taping it to the forehead (see Fig. 36-4). This keeps the tube from dislodging
when the patient moves but still allows it to pass into the intestine. Instead
of tape, a feeding tube attachment de-vice (Hollister) can be used to secure
the tube. This device ad-heres to the nose and uses an adjustable clip to hold
the tube in place (Fig. 36-5). After the nasoenteric tube has progressed into
the intestine (after approximately 24 hours), the tube may be taped in place.
Advancing the Nasoenteric Decompression Tube
After the tube has passed through the pyloric sphincter, it may be advanced 5 to 7.5 cm (2 to 3 in) every hour. To enable gravity and peristalsis to assist in the passage of the tube, the patient is generally asked to lie in the following positions in this order: on the right side for 2 hours, on the back for 2 hours, and then on the left side for 2 hours. Ambulation, if possible, also helps ad-vance the tube. If the tube is advanced too rapidly, it will curl and kink in the stomach. The tube is irrigated with normal saline so-lution every 6 to 8 hours to prevent blockage.
Monitoring the Patient and Maintaining Tube Function
If the
NG tube is used for decompression, it is attached to inter-mittent low suction.
If it is used for enteral nutrition, the end of the tube is plugged between
feedings. The nurse confirms tube placement before any fluids or medications are
instilled and once a shift for continuous feedings. Displacement of the tube
may be caused by tension on the tube (when the patient moves around in the bed
or room), coughing, tracheal or naso-tracheal suctioning, or airway intubation.
If the NG tube is re-moved inadvertently in a patient who has undergone
esophageal or gastric surgery, it is replaced by the physician, usually under
fluoroscopy to avoid trauma to the suture line.
It is
important to keep an accurate record of all fluid intake, feedings, and
irrigation. To maintain patency, the tube is irri-gated every 4 to 6 hours with
normal saline to avoid electrolyte loss through gastric drainage. If an
automatic flush enteral pump is used, the flushing schedule may be altered. The
nurse records the amount, color, and type of all drainage every 8 hours.
When
double- or triple-lumen tubes are used, each lumen is labeled according to its
intended use: aspiration, feeding, or bal-loon inflation. To avoid tension on
the tube, the portion of the tube from the nose to the drainage unit is fixed
in position, either with a safety pin or with adhesive tape loops that are
pinned to the patient’s pajamas or gown. The tube must be looped loosely to
prevent tension and dislodgement (see Fig. 36-4).
Providing Oral and Nasal Hygiene
Regular
and conscientious oral and nasal hygiene is a vital part of patient care,
because the tube causes discomfort and pressure and may be in place for several
days. Moistened cotton-tipped swabs can be used to clean the nose, followed by
cleansing with a water-soluble lubricant. Frequent mouth care is comforting for
the pa-tient. The nasal tape is changed every 2 to 3 days, and the nose is
inspected for skin irritation. If the nasal and pharyngeal mucosa
areexcessively dry, steam or cool vapor inhalations may be beneficial. Throat
lozenges, an ice collar, chewing gum, or sucking on hard candies (if
permitted), and frequent movement also assist in relieving patient discomfort.
These activities keep the mucous membranes moist and help prevent inflammation
of the parotid glands.
Monitoring and Managin Potential Complications
Patients
with NG or nasoenteric intubation are susceptible to a variety of problems,
including fluid volume deficit, pulmonary complications, and tube-related
irritations. These potential com-plications require careful ongoing assessment.
Symptoms
of fluid volume deficit include dry skin and mucous membranes, decreased
urinary output, lethargy, and decreased body temperature. Assessment of fluid
volume deficit involves maintaining an accurate record of intake and output.
This in-cludes measuring NG drainage, fluid instilled by irrigation of the NG
tube, water taken by mouth, vomitus, water administered with tube feedings, and
intravenous (IV) fluids. Laboratory values, particularly blood urea nitrogen
and creatinine, are monitored. The nurse assesses 24-hour fluid balance and
reports negative fluid balance, increased NG output, interruption of IV
therapy, or any other disturbance in fluid intake or output.
Pulmonary
complications from NG intubation occur because coughing and clearing of the
pharynx are impaired, because gas buildup can irritate the phrenic nerve, and
because tubes may become dislodged, retracting the distal end above the
esopha-gogastric sphincter. Medications (antacids, simethicone, and
metoclopramide) are administered to decrease potential prob-lems. Signs and
symptoms of complications include coughing during the administration of foods
or medications, difficulty clearing the airway, tachypnea, and fever. Assessment
includes regular auscultation of lung sounds and routine assessment of vital
signs. It is important to encourage the patient to cough and to take deep
breaths regularly. The nurse also carefully confirms the proper placement of
the tube before instilling any fluids or medications.
Irritation
of the mucous membranes is a common complica-tion of NG intubation. The
nostrils, oral mucosa, esophagus, and trachea are susceptible to irritation and
necrosis. Visible areas are inspected frequently, and the adequacy of hydration
is assessed. When providing oral hygiene, the nurse carefully inspects the
mucous membranes for signs of irritation or excessive dryness. The nurse
palpates the area around the parotid glands to detect any tenderness or
enlarged nodes, indicating parotitis, and ob-serves for any skin or mucous
membrane irritation or necrosis. In addition, it is important to assess the
patient for esophagitis and tracheitis; symptoms include sore throat and
hoarseness.
Removing the Tube
Before
removing a tube, the nurse may intermittently clamp and unclamp the NG tube for
a trial period of 24 hours to ensure that the patient does not experience
nausea, vomiting, or distention. Before the tube is removed, it is flushed with
10 mL of normal saline to ensure that it is free of debris and away from the
gastric lining; then the balloon (if present) is deflated. Gloves are worn to
remove the tube. The tube is withdrawn gently and slowly for 15 to 20 cm (6 to
8 in) until the tip reaches the esophagus; the remainder is withdrawn rapidly
from the nostril. A nasointestinal tube is withdrawn at intervals of 10 minutes
until the end reaches the esophagus. If the tube does not come out easily,
force should not be used, and the problem should be reported to the physician.
As the tube is withdrawn, it is concealed in a towel, because the sight of it
may be unpleasant to the patient. After the tube is re-moved, the nurse
provides oral hygiene.
Related Topics