Chapter: Organic Chemistry: Aldehydes and ketones
Nucleophilic addition - Aldehydes and ketones
NUCLEOPHILIC ADDITION
Key Notes
Definition
Nucleophilic
addition involves the addition of a nucleophile to an aldehyde or a ketone. The
nucleophile adds to the electrophilic carbonyl carbon.
Overview
Charged
nucleophiles undergo nucleophilic addition with an aldehyde or ketone to give a
charged intermediate which has to be treated with acid to give the final
product. Neutral nucleophiles require acid catalysis and fur- ther reactions
can take place after nucleophilic addition.
Definition
As the name of the reaction suggests, nucleophilic addition involves the addition of a nucleophile to a molecule. This is a distinctive reaction for ketones and aldehydes and the nucleophile will add to the electrophilic carbon atom of the carbonyl group.
The nucleophile can be a
negatively charged ion such as cyanide or hydride, or it can be a neutral
molecule such as water or alcohol.
Overview
In general, addition of charged nucleophiles
results in the formation of a charged intermediate (Fig. 1). The reaction stops at this stage and acid has to be added
tocomplete the reaction.
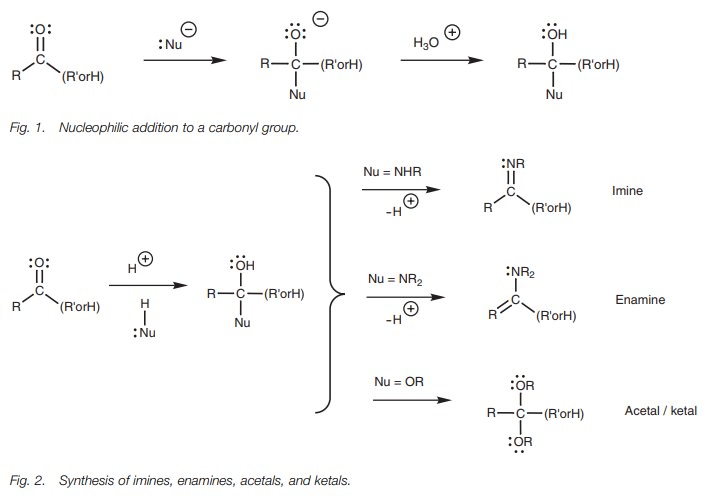
Neutral nucleophiles where nitrogen or oxygen
is the nucleophilic center are relatively
weak nucleophiles, and
an acid catalyst
is usually required.
After nucleophilic addition has occurred, further reactions may take
place leading to structures such as imines, enamines, acetals, and ketals.
Related Topics