Chapter: Clinical Anesthesiology: Clinical Pharmacology: Neuromuscular Blocking Agents
Nondepolarizing Muscle Relaxants: Unique Pharmacological Characteristics
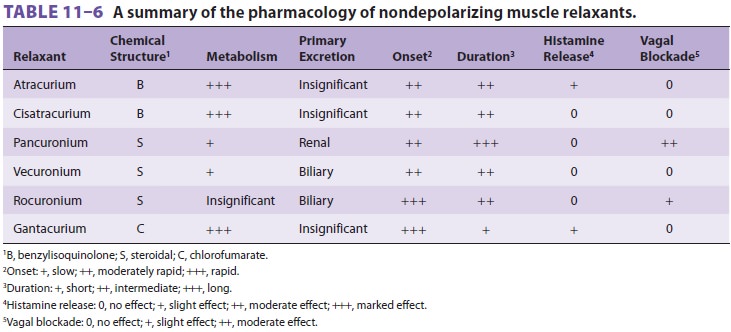
Nondepolarizing Muscle Relaxants
Unique Pharmacological Characteristics
In contrast to depolarizing muscle
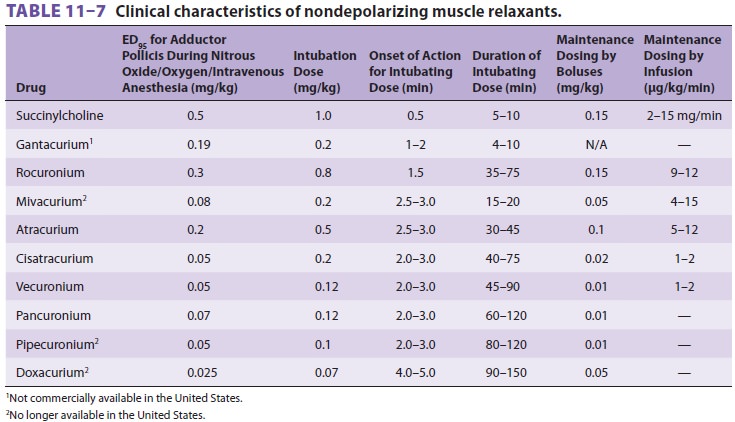
relaxants, there is a wide selection of nondepolarizing muscle relaxants ( Tables 11–6
and 11–7).
Based on their chemical structure, they can be classified as benzyl-isoquinolinium,
steroidal, or other compounds. It is often said that choice of a particular
drug depends on its unique characteristics, which are often related to its
structure; however, for most patients, the dif-ferences among the
intermediate-acting neuro-muscular blockers are inconsequential. In general,
steroidal compounds can be vagolytic, but this prop-erty is most notable with
pancuronium and clini-cally unimportant with vecuronium or rocuronium.
Benzylisoquinolines tend to release histamine. Because of structural
similarities, an allergic history to one muscle relaxant strongly suggests the
possi-bility of allergic reactions to other muscle relaxants, particularly
those in the same chemical class.
A. Suitability for Intubation
None of the currently available nondepolarizing muscle relaxants equals succinylcholine’s rapid onset of action or short duration. However, the onset of nondepolarizing relaxants can be quick-ened by using either a larger dose or a priming dose. The ED95 of any drug is the effective dose of a drug in 95% of individuals. For neuromuscular block-ers, one often specifies the dose that produces 95% twitch depression in 50% of individuals. One to two times the ED95 or twice the dose that produces 95% twitch depression is usually used for intubation. Although a larger intubating dose speeds onset, it exacerbates side effects and prolongs the duration of blockade. For example, a dose of 0.15 mg/kg of pancuronium may produce intubating condi-tions in 90 sec, but at the cost of more pronounced tachycardia—and a block that may be irreversible (by neostigmine) for more than 60 min. The conse-quence of a long duration of action is the ensuing difficulty in completely reversing the blockade and a subsequent increased incidence of postoperative pulmonary complications. As a general rule, the more potent the nondepolarizing muscle relax-ant, the slower its speed of onset; the “explana-tory dogma” is that greater potency necessitates a smaller dose, with fewer total drug molecules, which in turn, decreases the rate of drug binding opportunities at the neuromuscular junction.
The introduction of short- and
intermediate-acting agents has resulted in the greater use of priming doses.
Theoretically, giving 10% to 15% of the usual intubating dose 5 min before
induction will occupy enough receptors so that paralysis will quickly follow
when the balance of relaxant is administered. Use of a priming dose can produce
conditions suitable for intubation as soon as 60 sec following administra-tion
of rocuronium or 90 sec following administra-tion of other intermediate-acting
nondepolarizers. A priming dose does not usually
lead to clinically significant paralysis, which requires that 75% to 80% of the
receptors be blocked (a neuromuscular mar-gin of safety). In some patients,
however, the prim-ing dose produces distressing dyspnea, diplopia, or
dysphagia; in such instances, the patient should be reassured, and induction of
anesthesia should pro-ceed without delay. Priming can additionally cause
measureable deterioration in respiratory function (eg, decreased forced vital
capacity) and may lead to oxygen desaturation in patients with marginal
pul-monary reserve. These negative side effects are more common in older,
sicker patients.
Muscle groups vary in their sensitivity
to muscle relaxants. For example, the laryngeal muscles—whose relaxation is
important during intubation—recover from blockade more quickly than the
adductor pollicis, which is commonly mon-itored by the peripheral nerve
stimulator.
B. Suitability for Preventing Fasciculations
To prevent fasciculations and myalgias,
10% to 15% of a nondepolarizer intubating dose can be administered 5 min before
succinylcholine. When administered only shortly before succinylcholine,
myalgias, but not fasciculations, will be inhibited. Although most
nondepolarizers have been suc-cessfully used for this purpose, tubocurarine and
rocuronium have been most popular (precurari-zation); tubocurarine is no longer
available in the United States.
C. Maintenance Relaxation
Following intubation, muscle paralysis
may need to be maintained to facilitate surgery, (eg, abdominal operations), to
permit a reduced depth of anesthe-sia, or to control ventilation. There is
great variabil-ity among patients in response to muscle relaxants. Monitoring
neuromuscular function with a nerve stimulator helps to prevent over- and
underdos-ing and to reduce the likelihood of serious residual muscle paralysis
in the recovery room. Maintenance doses, whether by intermittent boluses or
con-tinuous infusion (Table 11–7), should be guided by the nerve stimulator and clinical signs (eg, spontaneous
respiratory efforts or movement). In some instances, clinical signs may precede
twitch recovery because of differing sensitivities to mus-cle relaxants between
muscle groups or technical problems with the nerve stimulator. Some return of
neuromuscular transmission should be evident prior to administering each
maintenance dose, if the patient needs to resume spontaneous ventila-tion at
the end of the anesthetic. When an infusion is used for maintenance, the rate
should be adjusted at or just above the rate that allows some return of
neuromuscular transmission so that drug effects can be monitored.
D. Potentiation by Inhalational Anesthetics
Volatile agents decrease nondepolarizer
dosage requirements by at least 15%. The actual degree of this postsynaptic
augmentation depends on both theinhalational anesthetic (desflurane > sevoflurane > isoflurane and enflurane > halothane >
N2O/O2/
narcotic) and the muscle relaxant employed (pan-curonium > vecuronium and atracurium).
E. Potentiation by Other Nondepolarizers
Some combinations of nondepolarizers
produce a greater than additive (synergistic) neuromuscular blockade. The lack
of synergism (ie, the drugs are only additive) by closely related compounds
(eg, vecuronium and pancuronium) lends credence to the theory that synergism
results from slightly dif-fering mechanisms of action.
F.
Autonomic Side Effects
In clinical doses, the nondepolarizers
differ in their relative effects on nicotinic and muscarinic choliner-gic
receptors. Some older agents (tubocurarine and, to a lesser extent, metocurine)
blocked autonomic ganglia, reducing the ability of the sympathetic ner-vous
system to increase heart contractility and rate in response to hypotension and
other intraoperative stresses. In contrast, pancuronium (and gallamine) block
vagal muscarinic receptors in the sinoatrial node, resulting in tachycardia.
All newer nondepo-larizing relaxants, including atracurium, cisatracu-rium,
vecuronium, and rocuronium, are devoid of significant autonomic effects in
their recommended dosage ranges
G. Histamine Release
Histamine release from mast cells can
result in bronchospasm, skin flushing, and hypotension from peripheral
vasodilation. Both atracurium and miva-curium are capable of triggering
histamine release, particularly at higher doses. Slow injection rates and H1 and H2 antihistamine pretreatment ameliorate these
side effects.
H. Hepatic Clearance
Only pancuronium and vecuronium are
metab-olized to any significant degree by the liver. Active metabolites likely
contribute to their clini-cal effect. Vecuronium and rocuronium depend heavily
on biliary excretion. Clinically, liver failure prolongs pancuronium and
rocuronium block-ade, with less effect on vecuronium, and no effect on
pipecuronium. Atracurium, cisatracurium, and mivacurium, although extensively
metabolized, depend on extrahepatic mechanisms. Severe liver disease does not
significantly affect clearance of atra-curium or cisatracurium, but the
associated decrease in pseudocholinesterase levels may slow the metabo-lism of
mivacurium.
I. Renal Excretion
Doxacurium, pancuronium, vecuronium, and
pipecuronium are partially excreted by thekidneys, and their action is
prolonged in patients with renal failure. The elimination of atracurium,
cisatracurium, mivacurium, and rocuronium is independent of kidney function.
Related Topics