Chapter: Biotechnology Applying the Genetic Revolution: Inherited Defects
Muscular Dystrophy
MUSCULAR DYSTROPHY
There are several forms of muscular dystrophy. These diseases
result in the wasting away of muscle tissue and cause premature death, usually
in the late teens or early 20s. There is no known cure. The most common form, Duchenne’s muscular dystrophy, is due
to defects in the protein dystrophin,
which plays a role in attaching the internal muscle fibrils to the membranes of
muscle cells (Fig. 16.11).
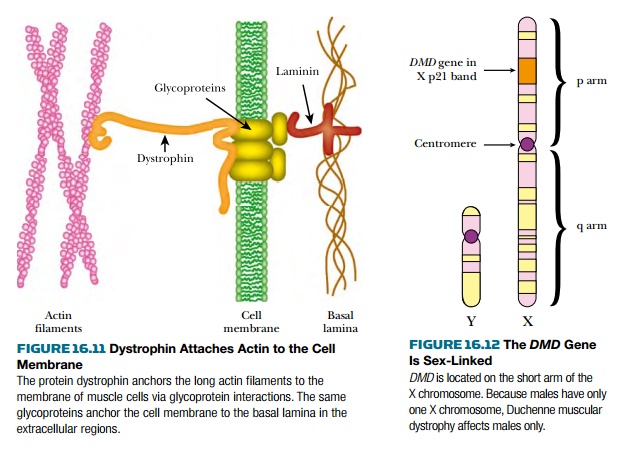
The DMD gene, responsible for this disease, is sex-linked and is located in the Xp21 band, close to the middle of the short (p) arm of the X chromosome (Fig. 16.12). Because the Y chromosome is shorter than the X chromosome, many genes that are present on the X chromosome do not have a corresponding partner on the Y chromosome. Therefore, females have two copies of the DMD gene, whereas males only have a single copy.
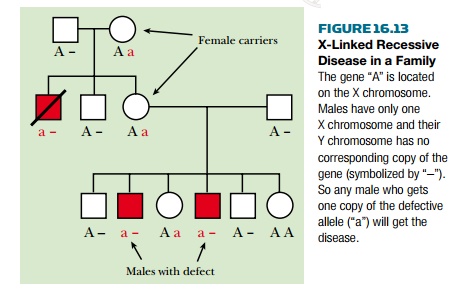
If the single copy of a sex-linked gene present in a male is defective, there is no backup copy and severe symptoms may result. In contrast, females with just one defective copy will usually have no symptoms, but they will be carriers and half of their male children will get the disease. The result is a pattern of inheritance in which only the male members of a family suffer from the disease. Figure 16.13 shows a family with several occurrences of an X-linked recessive disease. About two-thirds of Duchenne’s muscular dystrophy patients inherit the disease from their mothers and the other third get it as the result of new mutations, which arise at a frequency of approximately 1 in 10,000 gametes.
The DMD gene is even odder
than the cystic fibrosis gene. It takes up about 1.5% of the length of the X
chromosome and is longer than the entire genomes of some bacteria. The DMD gene
has roughly 75 exons and more than 2 million base pairs of DNA, of which less
than 1% are used to encode the protein. Despite this the encoded protein,
dystrophin, is gigantic. It has 4000 amino acids, so it is roughly 10 times as
large as an average protein. Such huge coding sequences are bigger targets for
detrimental mutations, which explains why the mutation rate for the DMD gene is
more than 10 times greater than for typical human genes. In most victims the
defect is due to alterations in just one or a few bases of the DMD gene.
However, about 10% of the victims have a deletion of DNA, which includes all or
part of the DMD gene.
One Duchenne’s muscular
dystrophy patient had a deletion large enough to locate to the Xp21 region of
the X chromosome using a light microscope. Using the subtractive hybridization
method, DNA from this patient was used to clone the DNA that was missing in the
deletion. In this approach, the DMD
gene to be cloned is present in the DNA from a healthy person. The sample of
DNA with the DMD deletion is used to
remove other, unwanted, genes from the healthy DNA, by hybridization. This
leaves behind the healthy copy of the DMD
gene, which is then cloned onto a suitable vector for further analysis.
Related Topics