Chapter: Biochemistry: The Importance of Energy Changes and Electron Transfer in Metabolism
Living Things Are Unique Thermodynamic Systems
Living Things
Are Unique Thermodynamic Systems
Questions arise frequently about whether living organisms obey the
laws of thermodynamics. The short answer is that they most deÞnitely do. Most
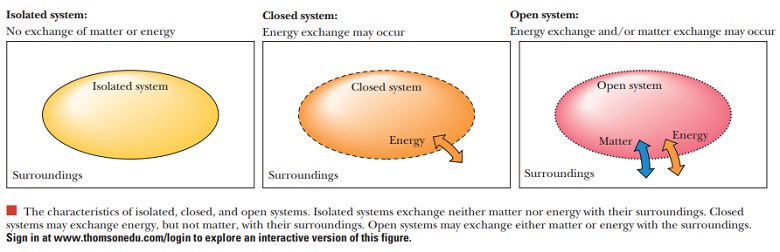
classical treatments of thermodynamics deal with closed systems at equilibrium.
A closed system can exchange energy, but not matter, with its surroundings.

A living organism is obviously not a closed system, but an open
system that can exchange both matter and energy with its surroundings. Because
living organisms are open systems, they cannot be at equilibrium as long as
they are alive, as shown in the following figure. They can, however, achieve a steady state, which is a stable
condition. It is the state in which living things can operate at maximum
thermodynamic efficiency. This point was established by Ilya Prigogine, winner
of the 1977 Nobel Prize in chemistry for his work on nonequilibrium
thermodynamics. He showed that for systems not at equilibrium, ordered structures
can arise from disordered ones. This treatment of thermodynam-ics is quite
advanced and highly mathematical, but the results are more directly applicable
to biological systems than those of classical thermodynamics. This approach
applies not only to liv-ing organisms but to the growth of cities and to
predictions of auto traffic.
Related Topics