Chapter: Basic & Clinical Pharmacology : Vasoactive Peptides
Inhibition of the Renin-Angiotensin System
INHIBITION OF THE
RENIN-ANGIOTENSIN SYSTEM
In
view of the importance of the renin-angiotensin system in car-diovascular
disease, considerable effort has been directed to developing drugs that
inhibit the system. A wide variety of agents that block the formation or action
of ANG II is now available. Some of these drugs block renin release, but most
inhibit the conversion of ANG I to ANG II, block angiotensin AT1
receptors, or inhibit the enzymatic action of renin.
Drugs That Block Renin Release
Several
drugs that interfere with the sympathetic nervous system inhibit the release of
renin. Examples are clonidine and propranolol. Clonidine inhibits renin release
by causing a centrally mediated reduction in renal sympathetic nerve activity,
and it may also exert a direct intrarenal action. Propranolol and other β-adrenoceptor–
blocking drugs act by blocking the intrarenal and extrarenal β recep-tors involved
in the neural control of renin release.
Angiotensin-Converting Enzyme Inhibitors
An
important class of orally active ACE inhibitors, directed against the active
site of ACE, is now extensively used. Captopril
and enalapril are examples of
the many potent ACEinhibitors that are available. These drugs differ in their
structure and pharmacokinetics, but they are interchangeable in clinical use.
ACE inhibitors decrease systemic vascular resistance without increasing heart
rate, and they promote natriuresis. As described, they are effective in the
treatment of hypertension, decrease morbidity and mortality in heart failure
and left ventricular dysfunction after myocardial infarction, and delay the
progression of diabetic nephropathy.
ACE
inhibitors not only block the conversion of ANG I to ANG II but also inhibit
the degradation of other substances, including bradykinin, substance P, and
enkephalins. The action of ACE inhibitors to inhibit bradykinin metabolism
contributes significantly to their hypotensive action (see Figure 11–5) and is
apparently responsible for some adverse side effects, including cough and
angioedema. These drugs are contraindicated in preg-nancy because they cause
fetal kidney damage.
Angiotensin Receptor Blockers
Potent
peptide antagonists of the action of ANG II are available for research use. The
best-known of these is the partial agonist, saralasin. Saralasin lowers blood pressure in hypertensive
patientsbut may elicit pressor responses, particularly when circulating ANG II
levels are low. Because it must be administered intrave-nously, saralasin is
used only for investigation of renin-dependent hypertension and other
hyperreninemic states.
The
nonpeptide ANG II receptor blockers
(ARBs) are of much greater interest. Losartan,
valsartan, eprosartan, irbesartan,candesartan, olmesartan, and telmisartan are orally active,potent,
and specific competitive antagonists of angiotensin AT1 receptors.
The efficacy of these drugs in hypertension is similar to that of ACE
inhibitors, but they are associated with a lower inci-dence of cough. Like ACE
inhibitors, ARBs slow the progression of diabetic nephropathy and valsartan has
been reported to decrease the incidence of diabetes in patients with impaired
glu-cose tolerance. The antagonists are also effective in the treatment of
heart failure and provide a useful alternative when ACE inhibi-tors are not
well tolerated. ARBs are generally well tolerated but should not be used by
patients with nondiabetic renal disease or in pregnancy Marfan syndrome is a
connective tissue disorder associated with aortic disease and other
abnormalities involving increased transforming growth factor (TGF)-β signaling. Since ANG
II increases TGF-β
levels, it was reasoned that blockade of the renin-angiotensin system might be
beneficial in Marfan syndrome. Promising initial results have been obtained
with losartan, and clinical trials are underway.
The
currently available ARBs are selective for the AT1 receptor. Since
prolonged treatment with the drugs disinhibits renin release and increases
circulating ANG II levels, there may be increased stimulation of AT2
receptors. This may be significant in view of the evidence that activation of
the AT2 receptor causes vasodila-tion and other beneficial effects.
AT2 receptor antagonists such as PD 123177 are available for
research but have no clinical applica-tions at this time. However, a selective
AT2agonist, compound 21,
lowers blood pressure in hypertensive animals and may be benefi-cial in human
hypertension. The clinical benefits of ARBs are similar to those of ACE
inhibitors, and it is not clear if one group of drugs has significant
advantages over the other. Combination therapy with an ACE inhibitor plus an
ARB has a number of potential advantages and is currently being investigated.
Renin Inhibitors
Cleavage
of angiotensinogen by renin (Figures 17–1 and 17–3) is the rate-limiting step
in the formation of ANG II and thus repre-sents a logical target for inhibition
of the renin-angiotensin system. Drugs that inhibit renin have been available
for many years but have been limited by low potency, poor bioavailability, and
short dura-tion of action. However, a new class of nonpeptide,
low-molecular-weight, orally active inhibitors has recently been developed.
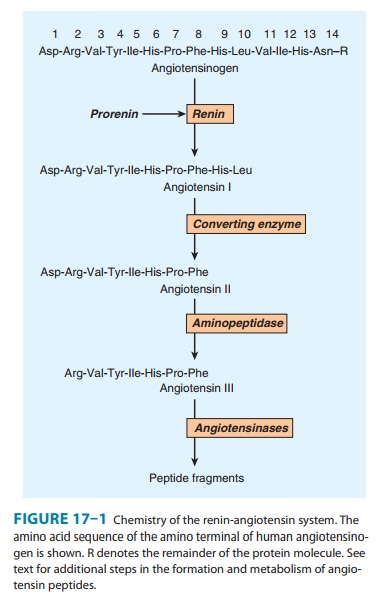
Aliskiren is the most advanced of these and the first to
beapproved for the treatment of hypertension. In healthy subjects, aliskiren
produces a dose-dependent reduction in plasma renin activity and ANG I and II
and aldosterone concentrations. In patients with hypertension, many of whom
have elevated plasma renin levels, aliskiren suppresses plasma renin activity
and causes dose-related reductions in blood pressure similar to those pro-duced
by ACE inhibitors and ARBs. The safety and tolerability of aliskiren appear to
be comparable to angiotensin antagonists and placebo. Aliskiren is
contraindicated in pregnancy.
Inhibition
of the renin-angiotensin system with ACE inhibitors or ARBs may be incomplete
because the drugs disrupt the negative feedback action of ANG II on renin
release and thereby increase plasma renin activity. Other antihypertensive
drugs, notably hydro-chlorothiazide and other diuretics, also increase plasma
renin activ-ity. Aliskiren not only decreases baseline plasma renin activity in
hypertensive subjects but also eliminates the rise produced by ACE inhibitors,
ARBs, and diuretics and thereby results in a greater anti-hypertensive effect.
Renin inhibition has thus proved to be an important new approach to the
treatment of hypertension.
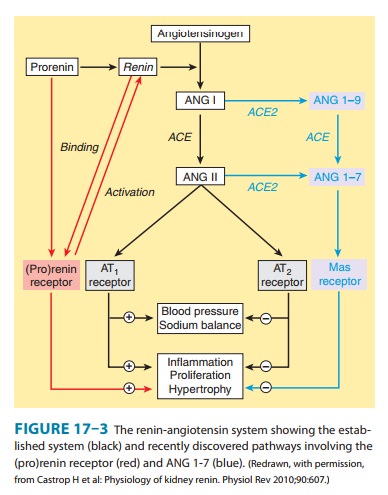
Prorenin Receptors
For
many years, prorenin was considered to be an inactive pre-cursor of renin, with
no function of its own. Thus the observationnoted above in the section on renin
that prorenin circulates at high levels was surprising. Recently, however, a
receptor that preferentially binds prorenin has been identified. Since it also
binds active renin, the receptor is referred to as the (pro)renin receptor.
The
receptor is a 350-amino acid protein with a single trans-membrane domain. When
prorenin binds to the (pro)renin receptor, it undergoes a conformational change
and becomes fully active. The catalytic activity of active renin also increases
further when it binds to the receptor. The activated prorenin and renin
interact with circulating angiotensinogen to form angio-tensin (Figure 17–3).
However, binding of prorenin to the recep-tor also activates intracellular
signaling pathways that differ depending on the cell type. For example, in
mesangial and vas-cular smooth muscle cells, prorenin binding activates MAP
kinases and expression of profibrotic molecules. Thus, elevated prorenin levels
(as occur, for example, in diabetes mellitus) could produce a variety of adverse
effects via both angiotensin-depen-dent and independent pathways. Recent
research indicates that the (pro)renin receptor is functionally linked to the
vacuolar proton-ATPase (ATP6ap2) and is necessary for Wnt signaling pathways
involved (independently of renin) in stem cell biology, embryology, and cancer.
A
synthetic peptide named handle region peptide (HRP), which consists of the
amino acid sequence corresponding to the “handle” region of the prorenin
prosegment, has been synthesized and shown to competitively inhibit binding of
prorenin to the (pro)renin receptor. HRP has beneficial effects in the kidneys
of diabetic rats and there is considerable inter-est in developing
noncompetitive antagonists of the (pro)renin receptor.
This
novel receptor could be important in cardiovascular and other diseases, but at
the present time its role in human pathology is far from clear.
Related Topics