Chapter: Basic & Clinical Pharmacology : Vasoactive Peptides
Endothelins
ENDOTHELINS
The
endothelium is the source of a variety of substances with vasodilator (PGI2
and nitric oxide) and vasoconstrictor activities. The latter include the
endothelin family, potent vasoconstrictor peptides that were first isolated
from aortic endothelial cells.
Biosynthesis, Structure, & Clearance
Three
isoforms of endothelin have been identified: the originally described
endothelin, ET-1, and two similar
peptides, ET-2 and ET-3. Each isoform is a product of a
different gene and is synthe-sized as a prepro form that is processed to a
propeptide and then to the mature peptide. Processing to the mature peptides
occurs through the action of endothelin-converting enzyme. Each endothelin is a
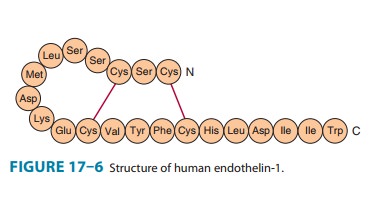
21-amino-acid peptide containing two disulfide bridges. The structure of ET-1 is
shown in Figure 17–6.
Endothelins
are widely distributed in the body. ET-1 is the predominant endothelin secreted
by the vascular endothelium. It is also produced by neurons and astrocytes in
the central nervous system and in endometrial, renal mesangial, Sertoli, breast
epithe-lial, and other cells. ET-2 is produced predominantly in the kid-neys
and intestine, whereas ET-3 is found in highest concentration
in
the brain but is also present in the gastrointestinal tract, lungs, and
kidneys. Endothelins are present in the blood but in low con-centration; they
apparently act locally in a paracrine or autocrine fashion rather than as
circulating hormones.
The
expression of the ET-1 gene is increased by growth factors and cytokines,
including transforming growth factor-β (TGF-β) and interleukin 1 (IL-1), vasoactive
substances including ANG II and AVP, and mechanical stress. Expression is
inhibited by nitric oxide, prostacyclin, and ANP.
Clearance
of endothelins from the circulation is rapid and involves both enzymatic degradation
by NEP 24.11 and clearance by the ETB receptor.
Actions
Endothelins
exert widespread actions in the body. In particular, they cause potent
dose-dependent vasoconstriction in most vascu-lar beds. Intravenous
administration of ET-1 causes a rapid and transient decrease in arterial blood
pressure followed by a sus-tained increase. The depressor response results from
release of prostacyclin and nitric oxide from the vascular endothelium, whereas
the pressor response is due to direct contraction of vascu-lar smooth muscle.
Endothelins also exert direct positive inotropic and chronotropic actions on
the heart and are potent coronary vasoconstrictors. They act on the kidneys to
cause vasoconstric-tion and decrease glomerular filtration rate and sodium and
water excretion. In the respiratory system, they cause potent contraction of
tracheal and bronchial smooth muscle. Endothelins interact with several
endocrine systems, increasing the secretion of renin, aldosterone, AVP, and
ANP. They exert a variety of actions on the central and peripheral nervous
systems, the gastrointestinal sys-tem, the liver, the urinary tract, the male
and female reproductive systems, the eye, the skeletal system, and the skin.
Finally, ET-1 is a potent mitogen for vascular smooth muscle cells, cardiac
myo-cytes, and glomerular mesangial cells.
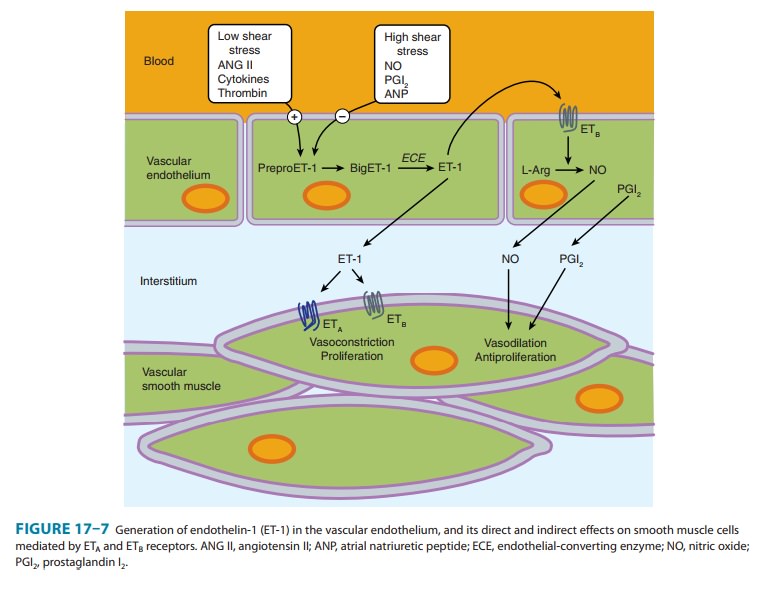
Endothelin
receptors are widespread in the body. Two endothe-lin receptor subtypes, termed
ETA and ETB, have been cloned and
sequenced. ETA receptors have a high affinity for ET-1 and a low affinity
for ET-3 and are located on smooth muscle cells, where they mediate
vasoconstriction (Figure 17–7). ETB receptors have approximately
equal affinities for ET-1 and ET-3 and are primar-ily located on vascular
endothelial cells, where they mediate release of PGI2 and nitric
oxide. Some ETB receptors are also present on smooth muscle cells
and mediate vasoconstriction. Both receptor subtypes belong to the G
protein-coupled seven-transmembrane domain family of receptors.
The
signal transduction mechanisms triggered by binding of ET-1 to its vascular
receptors include stimulation of phospholipase C, formation of inositol
trisphosphate, and release of calcium from the endoplasmic reticulum, which
results in vasoconstriction. Conversely, stimulation of PGI2 and
nitric oxide synthesis results in decreased intracellular calcium concentration
and vasodilation.
Related Topics