Chapter: Pharmaceutical Drug Analysis: Infrared Spectrophotometry
Infrared Spectrophotometry
INFRARED SPECTROPHOTOMETRY
INTRODUCTION
The infrared
spectrum provides the largest number of characteristic properties of a
compound. It also serves as a powerful ‘analytical
tool’ for the extensive and intensive study of molecular structure.
In fact, infrared
absorption spectra are due to changes in vibrational energy accompanied by
changes in rotational energy. Broadly speaking, the range in the
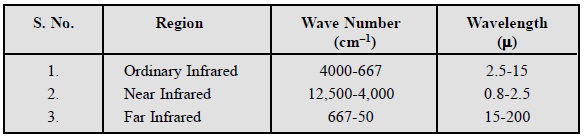
electromagnetic spectrum that extends from 0.8 to 200 μ is referred to as the infrared region. In usual
practice, however, either the wavelength (μ) or the wave number (cm–1) is employed to
measure the position of a given infrared absorption. More precisely, the
infra-red regions may be categorized into three distinct zones based on their
respective wave numbers and wave-lengths as stated below :
Another school of thought advocates that there are two
general regions in the infrared spectrum, namely : (a) Group frequency region :
having a wavelength ranging from 2.5 to 8.0 μ and a wave number from 4000-1300 cm–1 ; (b) Fingerprint
region : having a wavelength ranging from 8.0-2.5 μ and a wave number from 1300-400 cm–1.
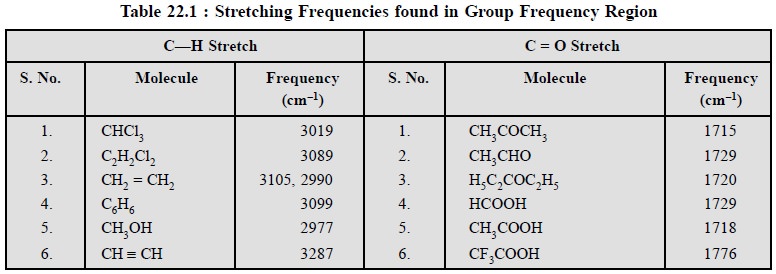
1. GROUP FREQUENCY REGION
Here, the stretching and bending vibrational bands associated with specific structural or functional groups are observed frequently.
Example : The C = O stretching
frequency is about 1700 cm–1 ; whereas the C—H stretching frequency
is about 3000 cm–1 and both of them are almost independent of the
rest of the molecule as depicted in Table 22.1.
2. FINGERPRINT REGION
Here, the vibrational modes depend solely and strongly on
the rest of the molecule.
Example : The C—C stretching frequency
depends largely on what else is bonded to the carbon atoms.
It is interesting to observe here that this particular
region of the spectrum is densely populated with bands. As we know that no two ‘fingerprints’ could be identical in
human beings, exactly in a similar manner no two compounds may have the same
‘fingerprint region’. Thus, each and every molecule essen-tially gives rise to
a unique spectrum which offers a characteristic feature of the same.
Related Topics