Chapter: 11th Biochemistry : Chapter 5 : Carbohydrates
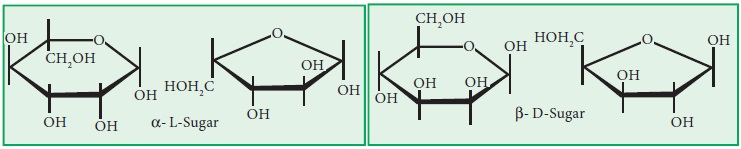
Haworth's Projection formula
Haworth’s Projection formula
A
Haworth Projection is a common way of writing a structural formula to represent
the cyclic structure of monosaccharides. It was named in the remembrance of
chemist Sir Norman Haworth. Let’s consider two sugars, one an aldohexose, the
other an aldopentose. Fischer projections are shown below. Recall the carbon
with the star (*) next to it is the one that determines if the sugar is a D
-sugar or an L-sugar.
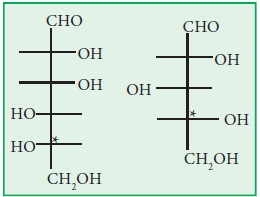
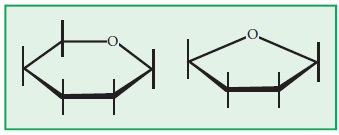
The Following are the rules.
1. Draw
the basic structure for the sugar.
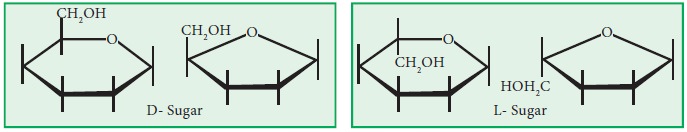
2. If
the sugar is a D-sugar, place a –CH2OH above the ring on the carbon
to the left of the oxygen, for an L-sugar place it below the ring.
3. For
an -sugar place an ^- OH below the ring on the carbon to the right of the ring
oxygen, for an β-sugar place the - OH above the ring.
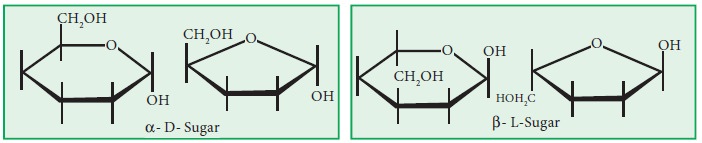
4. Finally, –OH groups on the right
go below the ring and those on the left above, using the–CH2OH group
as the reference point for both projections.
Related Topics