Chapter: Medical Microbiology: An Introduction to Infectious Diseases: Bacterial Processes
Growth of Bacterial Cultures
GROWTH OF BACTERIAL CULTURES
Solutions of nutrients that support the growth of bacteria are called media (singular, medium), which can be solidified by the incorporation of agar. The introduction of livecells into liquid sterile media or onto the surface of solidified media is calledinoculation. A population of bacterial cells is referred to as a culture. If the population is genetically homogeneous (ie, if all cells belong to the same strain of the same species), it is called a pure culture. Study of bacteria usually requires pure cultures, which can be obtained inseveral ways. The most common is to spread a very dilute suspension of a mixed culture on the surface of medium solidified with agar. Growth of individual cells deposited across the surface of solidified medium leads to visible mounds of bacterial mass called colonies. The cells in a colony are usually descended from a single original cell and, inthis case, constitute a clone. There is little difference between a pure culture and a clone, except that a pure culture may have been produced by the original inoculation of several identical cells. Colonies of different species and strains show marked differences in size, form, and consistency resulting from differences in growth rates, surface properties of the organisms, and their response to the gradients of nutrients and metabolites that develop within the colony as it enlarges. This facilitates subculturing to pure cultures.
Growth of a liquid bacterial culture can be monitored by removing samples at timed intervals and placing suitable dilutions in or on solidified medium to obtain a count of the number of colonies that develop. The count can be directly extrapolated to the number of viable units in the original sample (which, because certain bacteria clump or form chains, may not represent the number of bacterial cells). Growth can also be measured by determining the number of total cells in each sample. Direct count with a microscope is simple but tedious; more sensitive and accurate counts can be made with the aid of an electronic particle counter. More often, the turbidity of the culture is measured, because bacterial cultures above approximately 106 cells/mL are visibly turbid, and turbidity is proportional to the total mass of bacterial protoplasm present per milliliter. Turbidity is quickly and easily measured by means of a spectrophotometer.
The growth rate of a bacterial culture depends on three factors: the species of bac-terium, the chemical composition of the medium, and the temperature. The time needed for a culture to double its mass or cell number is in the range of 30 to 60 minutes for most pathogenic bacteria in rich media. Some species can double in 20 minutes (E. coli and re-lated organisms), and some (eg, some mycobacteria) take almost as long as mammalian cells, 20 hours. In general, the greater the variety of nutrients provided in the medium, the faster growth occurs. This superficially simple fact actually depends on the operation of metabolic regulatory devices of considerable sophistication, which, as we shall see in the next section, ensures that building blocks provided in the environment not be wastefully synthesized by the cells. For each bacterial species, there is a characteristic optimum tem-perature for growth, and a range, sometimes as broad as 40°, within which growth is pos-sible. Most pathogens of warm-blooded creatures have a temperature optimum for growth near normal body temperature, 37°C; growth often occurs at room temperature, but slowly. Therefore, incubators set at 35 to 37°C are used for culture of most clinical speci-mens. Exceptions to this rule include some organisms causing superficial infections for which 30°C is more suitable. As a group, bacteria have the widest span of possible growth temperatures, extending over the entire range of liquid water, 0°C to 100°C. Bac-teria that grow best at refrigerator temperatures are called psychrophiles, those that grow above 50°C are called thermophiles; in between are the mesophiles, including virtually all pathogens.
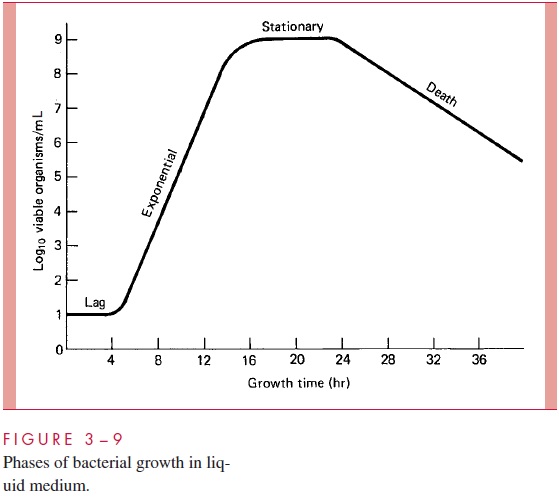
When first inoculated, liquid cultures of bacteria characteristically exhibit a lagperiod during which growth is not detectable. This is the first phase of what is called the culture growth cycle (Fig 3 – 9). During this lag, the cells are actually quite active in ad-justing the levels of vital cellular constituents necessary for growth in the new medium. Eventually net growth can be detected, and after a brief period of accelerating growth, the culture enters a phase of constant, maximal growth rate, called the exponential orlogarithmic phase of growth, during which the generation time is constant. During thisphase, cell number, and total cell mass, and amount of any given component of the cells increase at the same exponential rate; such growth is called balanced growth, or steady-state growth. The full reproductive potential of bacteria is exhibited during this phase:one cell gives rise to 2 cells in 1 generation, to 8 cells after 3 generations, to 1024 cells after 10 generations, and to about 1 million cells in 20 generations. For a bacterial species with a generation time of 20 minutes, therefore, it takes less than 7 hours in the exponen-tial phase of growth to produce a million cells from one.
By use of an equation for exponential growth, it can be demonstrated that 2 days of growth at this rate would be sufficient to generate a mass of bacteria equal to 500 times the mass of the earth. Fortunately, this never occurs, but not because the equation is faulty. Constant growth rate requires that there be no change in the supply of nutrients or the concentration of toxic by-products of metabolism (such as organic acids). This con-stancy can exist for only a short time (hours) in an ordinary culture vessel. Then growth becomes progressively limited (decelerating phase) and eventually stops (stationaryphase). Cells in the stationary phase are different from those in the exponential phase.They are smaller, have a different complement of enzymes (to deal with survival during starvation), and have fewer ribosomes per unit mass. When an inoculum of such cells is placed into fresh medium, exponential growth cannot resume immediately, and hence the lag period is observed. Note that there is no lag phase if the inoculum consists of exponential-phase cells. Prolonged incubation of a stationary-phase culture leads to cell death for many bacterial species (such as the pneumococcus), although many (such as E.coli) are hardy enough to remain viable for days. During thedeath phaseordeclineofa culture, cell viability is lost by exponential kinetics as described. As already noted, for those Gram-positive species that can sporulate, entry into the stationary phase usually triggers this event.
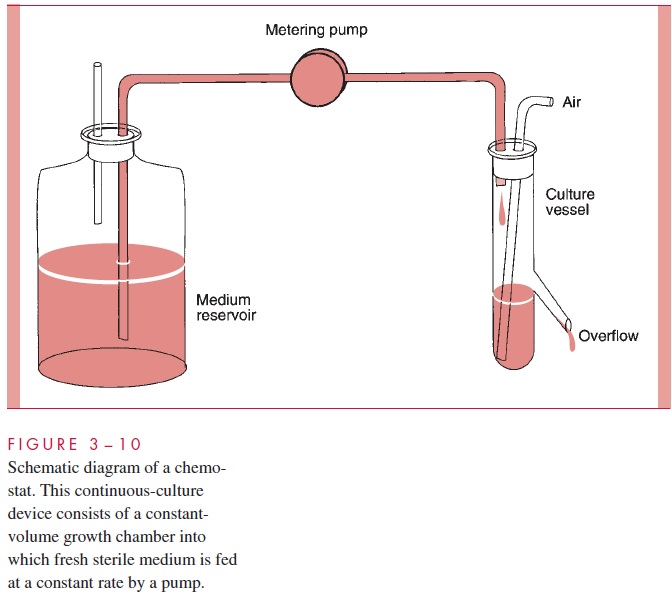
One way to maintain a culture in exponential, steady-state (balanced) growth for long periods is to use a device in which fresh medium is continuously added but the total vol-ume of culture is held constant by an overflow tube. One such constant-volume device is called a chemostat; it operates by infusing fresh medium containing a limiting nutrient at a constant rate, and the growth rate of the cells is set by the flow rate (Fig 3 – 10). A simi-lar constant-volume device is the turbidostat; it operates by the infusion of fresh medium by a pump controlled indirectly by the turbidity of the culture. Although such de-vices may sound artificial, they mimic many situations of interest to medical microbiolo-gists. Most of the places in which bacteria live on and within our bodies, in health and disease, provide conditions more closely resembling those of nutrient-limited continuous-culture devices than of enclosed flasks.
Related Topics