Chapter: Medical Microbiology: An Introduction to Infectious Diseases: Bacterial Processes
Fueling Reactions - Cell Growth of Bacterial
Fueling Reactions
Fueling reactions provide the cell with energy and with the 12 precursor metabolitesused in biosynthetic reactions (Fig 3 – 1).
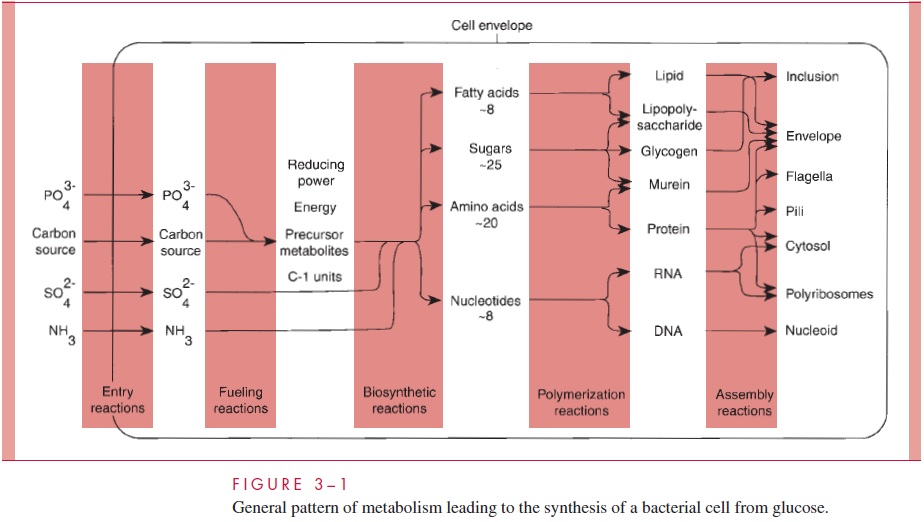
The first step is the capture of nutrients from the environment. Both Gram-positive and Gram-negative cells have surrounded themselves with envelopes designed in part to exclude potentially harmful substances and, therefore, have had to evolve a number of ways to ensure rapid transport of selected solute molecules through the envelope. Meth-ods used by Gram-negative cells are summarized in Figure 3 – 2.
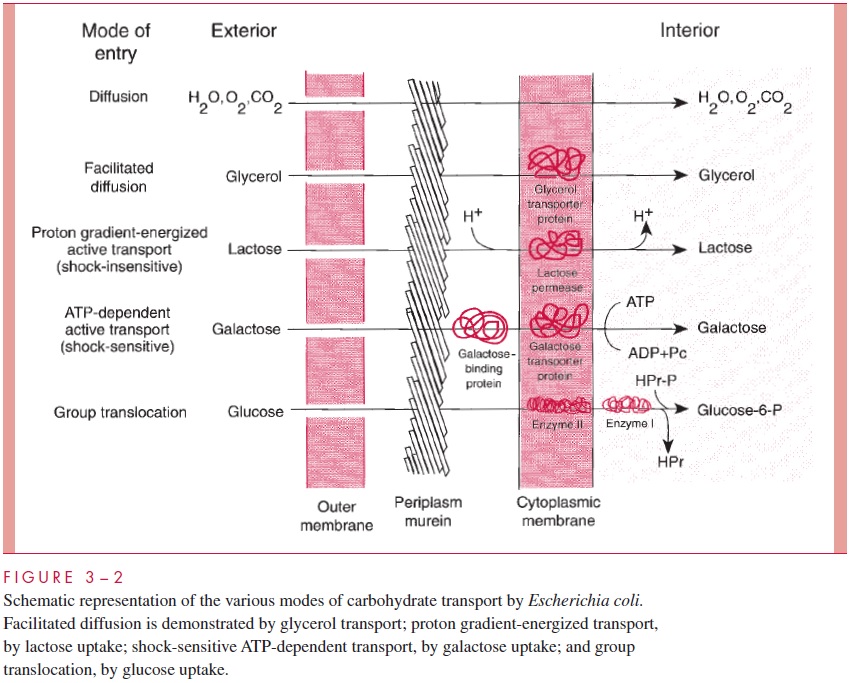
Almost no important nutrients enter the cell by simple diffusion, because the cell mem-brane is too effective a barrier to most molecules (the exceptions are carbon dioxide, oxy-gen, and water). Some transport occurs by facilitated diffusion in which a protein carrier in the cell membrane, specific for a given compound, participates in the shuttling of molecules of that substance from one side of the membrane to the other. Glycerol enters E. coli cells in this manner, and in bacteria that grow in the absence of oxygen (anaerobic bacteria, see be-low) it is reasonably common for some nutrients to enter the cell and for fermentation byproducts to leave the cell by facilitated diffusion. Because no energy is involved, this process can work only with, never against, a concentration gradient of the given solute.
Active transport, like facilitated diffusion, involves specific protein molecules as car-riers of particular solutes, but the process is energy linked and can therefore establish a concentration gradient (active transport can pump “uphill”). Active transport is the most common mechanism in aerobic bacteria. Gram-negative bacteria have two kinds of active transport systems. In one, called shock-sensitive because the working components can be released from the cell by osmotic shock treatments, solute molecules cross the outer membrane either by diffusion through the pores of the outer membrane (as in the case of galactose) or by a special protein carrier (as in the case of maltose). In the periplasm, the solute molecules bind to specific binding proteins, which interact with carrier proteins in the cell membrane. Shock-sensitive systems couple the transport across the cell mem-brane with the hydrolysis of ATP.
The other type of active transport involves only cell membrane components (and hence is shock insensitive) and is distinctive in that solute transport is coupled to the simultaneous passage of protons (H ) through the membrane. The energy for this type of active transport is therefore derived not from ATP hydrolysis but from the proton gradient set up by electron transport within the energized cell membrane.
Finally, group translocation is an extremely common means of transport in the ab-sence of oxygen. It involves the chemical conversion of the solute into another molecule as it is transported. The phosphotransferase system for sugar transport, which involves the phosphorylation of sugars such as glucose by specific enzymes, is a good example.
The transport of iron and other metal ions needed in small amounts for growth is special and of particular importance in virulence. There is little free Fe3 in human blood or other body fluids, because it is sequestered by iron-binding proteins (eg, trans-ferrin in blood andlactoferrin in secretions). Bacteria must have iron to grow, and theircolonization of the human host requires capture of iron. Bacteria secrete siderophores (iron-specific chelators) to trap Fe3 ; the iron-containing chelator is then transported into the bacterium by specific active transport. One example of a siderophore is aer-obactin (a citrate type of hydoxamate), another is enterobactin (a catechol). Somesiderophores are produced as a result of enzymes encoded not in the bacterial genome, but in the genome of a plasmid, providing another example of the many ways in which plasmids are involved in virulence.
Once inside the cell, sugar molecules or other sources of carbon and energy are metabolized by the Embden – Meyerhof glycolytic pathway, the pentose phosphate pathway, and the Krebs cycle to yield the carbon compounds needed for biosynthesis.
Some bacteria have central fueling pathways (eg, the Entner – Doudoroff pathway) other than those familiar in mammalian metabolism.
Working in concert, the central fueling pathways produce the 12 precursor metabo-lites. Connections to fermentation and respirationpathways allow the reoxidation of reduced coenzyme nicotinamide adenine dinucleotide (NADH) to NAD and the genera-tion of ATP. Bacteria make ATP by substrate phosphorylation in fermentation or by a combination of substrate phosphorylation and oxidative phosphorylation in respiration. (Photosynthetic bacteria are not important in medicine.)
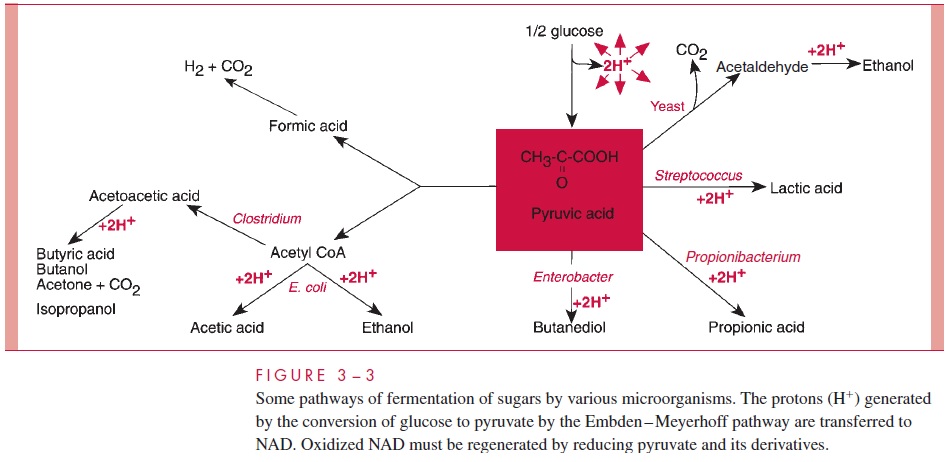
Fermentation is the transfer of electrons and protons via NAD directly to an or-ganic acceptor. Pyruvate occupies a pivotal role in fermentation (Fig 3 – 3). Fermentation is an inefficient way to generate ATP, and consequently huge amounts of sugar must be fermented to satisfy the growth requirements of bacteria anaerobically. Large amounts of organic acids and alcohols are produced in fermentation. Which compounds are produced depends on the particular pathway of fermentation employed by a given species, and therefore the profile of fermentation products is a diagnostic aid in the clinical laboratory.
Respiration involves fueling pathways in which substrate oxidation is coupled to thetransport of electrons through a chain of carriers to some ultimate acceptor, which is fre-quently, but not always, molecular oxygen. Other inorganic (eg, nitrate) as well as organic compounds (eg, succinate) can serve as the final electron acceptor, and therefore many or-ganisms that cannot ferment can live in the absence of oxygen (eg, Pseudomonas aerugi-nosa in the human colon).
Respiration is an efficient generator of ATP. Respiration in prokaryotes as in eukary-otes occurs by membrane-bound enzymes (quinones, cytochromes, and terminal oxidases), but in prokaryotes the cell membrane rather than mitochondrial membranes provide the physical site. The passage of electrons through the carriers is accompanied by the secretion from the cell of protons, generating an H differential between the external surface of the cytosol membrane and the cell interior. This differential, called the proton-motive force, can then be used to (1) drive transport of solutes by the shock-insensitive systems of active transport (see above); (2) power the flagellar motors that rotate the filaments and result in cell motility in the case of motile species; and (3) generate ATP by coupling the phospho-rylation of adenosine diphosphate (ADP) to the passage of protons inward through special channels in the cell membrane. The last pathway, facilitated by the enzyme anachronisti-cally called membrane ATPase, can in fact function in either direction, coupling ADP phosphorylation to the inward passage of protons down the gradient or hydrolyzing ATP to accomplish the secretion of protons to establish a proton-motive force. The latter process explains how cells can generate a proton-motive force anaerobically (i.e., in the absence of electron transport).
In evolving to colonize every conceivable nook and cranny on this planet, bacteria have developed distinctive responses to oxygen. Bacteria are conveniently classified ac-cording to their fermentative and respiratory activities but much more generally by their overall response to the presence of oxygen. The response depends on their genetic ability to ferment or respire but also on their ability to protect themselves from the deleterious effects of oxygen.
Oxygen, though itself only mildly toxic, gives rise to at least two extremely reactive and toxic substances, hydrogen peroxide (H2O2) and the superoxide anion (O2 ). Peroxide is produced by reactions (catalyzed by flavoprotein oxidases) in which elec-trons and protons are transferred to O2 as final acceptor. The superoxide radical is produced as an intermediate in most reactions that reduce molecular O2. Superoxide is partially detoxified by an enzyme, superoxide dismutase, found in all organisms (prokaryotes and eukaryotes) that survive the presence of oxygen. Superoxide dismutase catalyzes the reaction
2O-2+2H+ - - > H2O2+O2.
Hydrogen peroxide is degraded by peroxidases by the reaction
H2O2+H2A - - > 2H2O+A
where A is any of a number of chemical groups (in the case in which H2A is another mol-ecule of H2O2, the reaction yields 2H2O+O2, and the peroxidase is called catalase). Bacteria that lack the ability to make superoxide dismutase and catalase are exquisitely sensitive to the presence of molecular oxygen and, in general, must grow anaerobically using fermentation. Bacteria that possess these protective enzymes can grow in the pres-ence of oxygen, but whether they use the oxygen in metabolism or not depends on their ability to respire. Whether these oxygen-resistant bacteria can grow anaerobically de-pends on their ability to ferment.
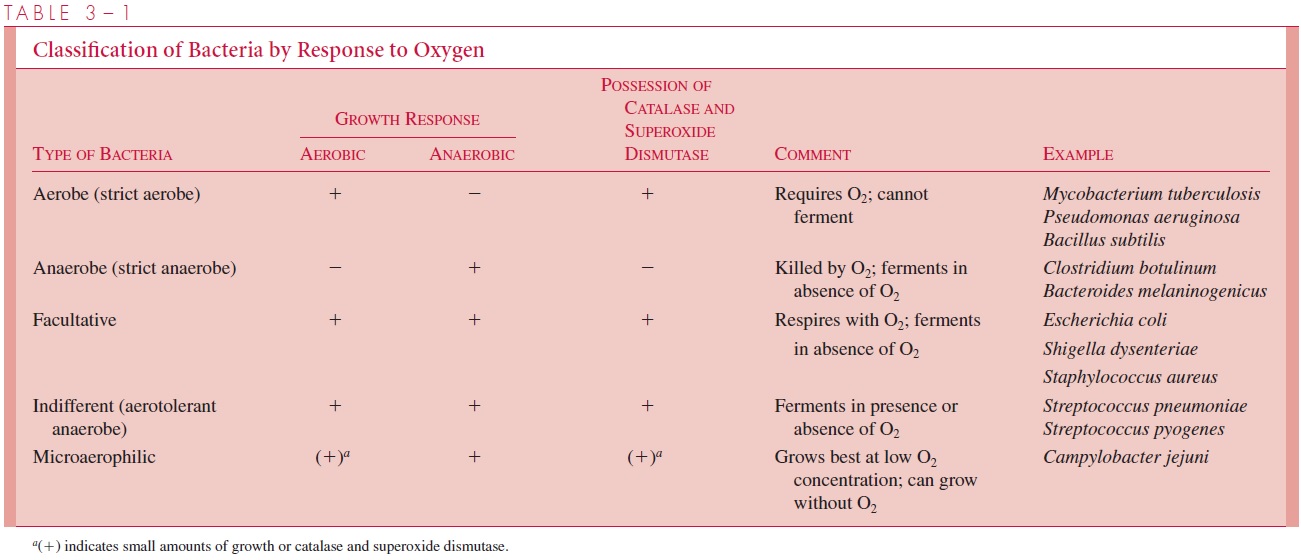
Various combinations of these two characteristics (oxygen resistance and the ability to use molecular oxygen as a final acceptor) are represented in different species of bacte-ria, resulting in the five general classes shown in Table 3 – 1. There are important pathogens within each class. Both the nature of the diseases they cause and the methods for cultivating and identifying these pathogens in the laboratory are dictated to a large ex-tent by their response to oxygen. Many medically important bacteria classified as anaer-obes (including those listed in Table 3 – 1) are in fact moderately aerotolerant, and may possess low levels of superoxide dismutases and peroxidases that provide some survival protection, if not the ability to grow.
Related Topics