Chapter: Genetics and Molecular Biology: Chemotaxis
Fundamental Properties of Chemotaxis
Fundamental Properties of Chemotaxis
The capillary tube assay permits several simple
measurements delimiting methods by which cells accomplish chemotaxis. The
first is that metabolism of attractant is not required for chemotaxis. An
analog of galactose, fucose, is not metabolized by Escherichia coli, and yet this serves as an attractant in the
capillary tube assay. Another line of evidence leading to this same conclusion
is that some mutants that are unable to metabolize galactose are still able to
swim toward galactose.
Since cells can swim toward or away from a wide
variety of chemi - cals, it is likely that they possess a number of receptors,
each with different specificity. Therefore the question arises as to how many
different types of receptors a cell possesses. This question can be answered by
a cross-inhibition test. Consider the case of fucose and galactose. Since their
structures are similar, it seems likely that cells detect both chemicals with
the same receptor protein. This conjecture
can be tested by using a high concentration of
fucose in both the tube and the drop so as to saturate the galactose receptor
and blind cells to the galactose that is placed only in the tube. Fucose does
blind cells to a galactose gradient, but it does not blind them to a serine
gradient. These findings prove that galactose and fucose share the same
receptor, but that serine uses a different receptor.
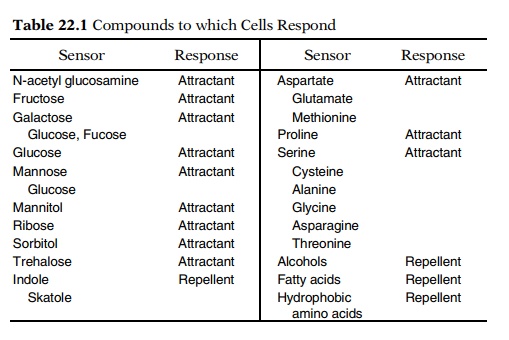
By blinding experiments and the use of mutants,
nine receptors have been found for sugars and three for amino acids (Table
22.1). The three amino acid receptors are not highly selective, and they allow
chemotaxis of E. coli toward 10 different amino acids. Except for the glucose
receptor, synthesis of the sugar receptors is inducible, as is the proline
The
capillary assay also allows a convenient determination of the sensitivity of
the receptors. By varying the concentration of attractant within the capillary,
the ranges over which the receptor will respond can be easily determined.
Typically, the lowest concentration is about 10-7 M, and the highest
concentration is about 10-1 or 10-2 M (Fig. 22.3). It is
not at all surprising that a detection system is no more sensitive than 10-7
M. First, at about 10-6 M the rate of diffusion of a sugar to a bacterium
is just adequate to support a 30-minute doubling time if every sugar molecule
reaching the cell is utilized. The ability to chemotact would not change this
lower limit by much.
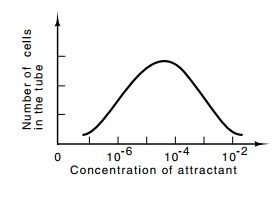
Figure
22.3 A typical re-sponse curve showing
the number of cells that have en-tered the capillary tube as a function of the
concentration of attractant placed in the tube.
The second limitation on detection sensitivity is the lowest concentration at which a sufficiently accurate measurement may be made in the one second measurement window set by the rate of random rotations. At a concentration of 10-7 M, the number of attractant molecules reaching the cell surface is
so low that statistical fluctuations in their number necessitate averaging for
a full second to obtain sufficient accuracy. Thus 10- 7 M is about
the lowest practical concentration to which bacteria can be expected to
chemotact.
The
actual receptor proteins for the different sugars have been sought, and several
have been identified biochemically. The receptors for galactose, maltose, and
ribose are found in the periplasm outside the inner membrane but inside the
peptidoglycan layer. These and other periplasmic proteins can be removed from
cells by osmotic shock.
No
periplasmic binding proteins have been found for the glucose, mannitol, and
trehalose systems. Instead, these systems use receptors that are tightly bound
to or located in the inner membrane. These receptors serve a double purpose as
they also function in the group translocation of their substrates into the cell
via the phosphotransferase transport system.
Related Topics