Chapter: Clinical Anesthesiology: Anesthetic Management: Respiratory Physiology& Anesthesia
Functional Respiratory Anatomy
Respiratory Physiology& Anesthesia
The importance of pulmonary physiology
to anes-thetic practice is obvious. The most commonly used anesthetics—the
inhalation agents—depend on the lungs for uptake and elimination. The most
important side effects of both inhalation and intravenously administered anesthetics
are pri-marily respiratory. Moreover, muscle paralysis, unusual positioning
during surgery, and techniques such as one-lung anesthesia and cardiopulmo-nary
bypass profoundly alter normal pulmonary physiology.
Much of modern anesthetic practice is
based on a thorough understanding of pulmonary physiology and may be considered
applied pulmonary physi-ology.
FUNCTIONAL RESPIRATORY ANATOMY
1. Rib Cage & Muscles of Respiration
The rib cage contains the two lungs,
each sur-rounded by its own pleura. The apex of the chest is small, allowing
only for entry of the trachea, esoph-agus, and blood vessels, whereas the base
is formed by the diaphragm. Contraction of the diaphragm— the principal
pulmonary muscle—causes the base of the thoracic cavity to descend 1.5–7 cm and
its contents (the lungs) to expand. Diaphragmatic movement normally accounts
for 75% of the change in chest volume. Accessory respiratory muscles also
increase chest volume (and lung expansion) by their action on the ribs. Each
rib (except for the last two) articulates posteriorly with a vertebra and is
angu-lated downward as it attaches anteriorly to the ster-num. Upward and
outward rib movement expands the chest.
During normal breathing, the diaphragm,
and, to a lesser extent, the external intercostal muscles are responsible for
inspiration; expiration is gener-ally passive. With increasing effort, the
sternoclei-domastoid, scalene, and pectoralis muscles can be recruited during
inspiration. The sternocleidomas-toid muscles assist in elevating the rib cage,
whereas the scalene muscles prevent inward displacement of the upper ribs
during inspiration. The pectoralis muscles can assist chest expansion when the
arms are placed on a fixed support. Expiration is normally passive in the
supine position, but becomes active in the upright position and with increased
effort. Exhalation may be facilitated by the abdominal muscles (rectus
abdominis, external and internal oblique, and transversus) and perhaps the
internal intercostal muscles—aiding the downward move-ment of the ribs.
Although not usually considered
respiratory muscles, some pharyngeal muscles are important in maintaining the
patency of the airway. Tonic and reflex inspiratory activity in the
genioglossus keeps the tongue away from the posterior pharyngeal wall. Tonic
activity in the levator palati, tensor palati, pal-atopharyngeus, and
palatoglossus prevents the soft palate from falling back against the posterior
phar-ynx, particularly in the supine position.
2. Tracheobronchial Tree
The trachea serves as a conduit for
ventilation and the clearance of tracheal and bronchial secretions. The trachea
begins at the lower border of the cricoid cartilage and extends to the level of
the carina and has an average length of 10–13 cm. It is composed of C-shaped
cartilaginous rings, which form the anterior and lateral walls of the trachea
and are connected posteriorly by the membranous wall of the trachea. The
external diameters of the trachea measure approximately 2.3 cm coronally and
1.8 cm sagitally in men, with corresponding values of 2.0 cm and 1.4 cm,
respectively, in women. The cricoid carti-lage is the narrowest part of the
trachea, with an aver-age diameter of 17 mm in men and 13 mm in women.
The trachea bifurcates at the carina
into the right and left main stem bronchi. The tracheal lumen narrows slightly
as it progresses toward the carina, with
the tracheal bifurcation located at the level of the sternal angle. The right
main stem bronchus lies in a more vertical orientation relative to the trachea,
whereas the left main stem bronchus lies in a more horizontal orientation. The
right main stem bron-chus continues as the bronchus intermedius after the
take-off of the right upper lobe bronchus. The distance from the tracheal
carina to the take-off of the right upper lobe bronchus is an average of 2.0 cm
in men and approximately 1.5 cm in women. One in every 250 individuals in the
general population may have an abnormal take-off of the right upper lobe
bronchus emerging from above the tracheal carina on the right side. The left
main stem bronchus is longer than the right main stem bronchus and mea-sures an
average of 5.0 cm in men and 4.5 cm in women. The left main stem bronchus
divides into the left upper lobe bronchus and the left lower lobe bronchus.
Humidification and filtering of inspired
air are functions of the upper airway (nose, mouth, and pharynx). The function
of the tracheobronchial tree is to conduct gas flow to and from the alveoli.
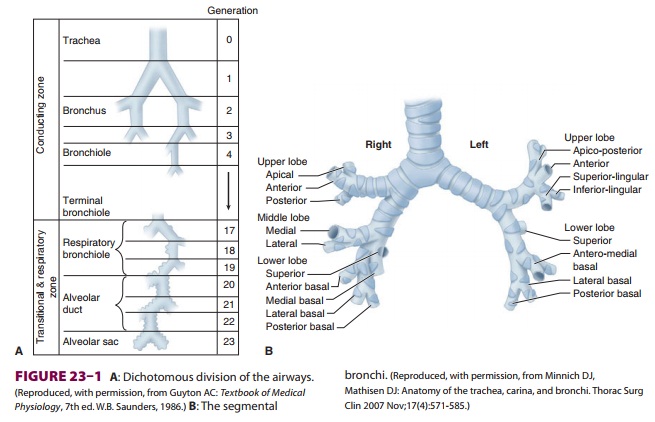
Dichotomous division (each branch dividing into two smaller branches), starting
with the trachea and ending in alveolar sacs, is estimated to involve 23
divisions, or generations (Figure 23–1). With each generation, the number
of airways is approximately doubled. Each alveolar sac contains, on average, 17
alveoli. An estimated 300 million alveoli provide an enormous membrane (50–100
m2) for gas exchange in the average adult.
With each successive division, the
mucosal epi-thelium and supporting structures of the airways gradually change.
The mucosa makes a gradual transition from ciliated columnar to cuboidal and
finally to flat alveolar epithelium. Gas exchange can occur only across the
flat epithelium, which begins to appear on respiratory bronchioles
(gen-erations 17–19). The wall of the airway gradually loses its cartilaginous
support (at the bronchioles) and then its smooth muscle. Loss of cartilaginous
support causes the patency of smaller airways to become dependent on radial
traction by the elas-tic recoil of the surrounding tissue; as a corollary,
airway diameter becomes dependent on total lung volume.
Cilia on the columnar and cuboidal
epithelium normally beat in a synchronized fashion, such that the mucus
produced by the secretory glands lining the airway (and any associated bacteria
or debris) moves up toward the mouth.
Alveoli
Alveolar size is a function of both
gravity and lung volume. The average diameter of an alveolus is thought to be
0.05–0.33 mm. In the upright posi-tion, the largest alveoli are at the pulmonary
apex, whereas the smallest tend to be at the base. With inspiration,
discrepancies in alveolar size diminish.
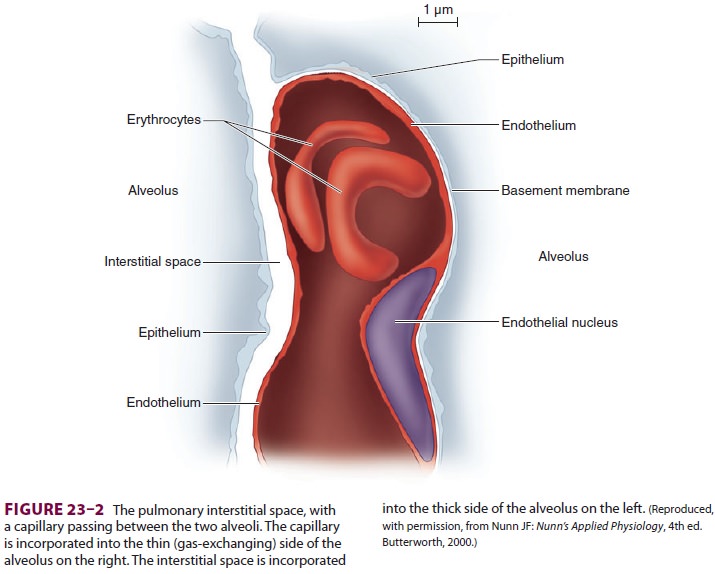
Each alveolus is in close contact with a
network of pulmonary capillaries. The walls of each alveolus are asymmetrically
arranged (Figure
23–2). On the thin side, where gas exchange occurs, the alveolar
epithelium and capillary endothelium are separated only by their respective
cellular and basement mem-branes; on the thick side, where fluid and solute
exchange occurs, the pulmonary interstitial spaceseparates alveolar epithelium
from capillary endo-thelium. The pulmonary interstitial space contains mainly
elastin, collagen, and perhaps nerve fibers. Gas exchange occurs primarily on
the thin side of the alveolocapillary membrane, which is less than 0.4 μm thick. The thick side (1–2 μm) provides structural support for the
alveolus.
The pulmonary epithelium contains at
least two cell types. Type I pneumocytes are flat and form tight (1-nm)
junctions with one another. These tight junctions are important in preventing
the passage of large oncotically active molecules such as albu-min into the
alveolus. Type II pneumocytes, which are more numerous than type I pneumocytes
(but because of their shape occupy less than 10% of the alveolar space), are round
cells that contain promi-nent cytoplasmic inclusions (lamellar bodies). These
inclusions contain surfactant, an important sub-stance necessary for normal
pulmonary mechanics . Unlike type I cells, type II pneumocytes are capable of
cell division and can produce type I
pneumocytes if the latter are destroyed.
They are also resistant to O2 toxicity.
Other cell types present in the lower
airways include pulmonary alveolar macrophages, mast cells, lymphocytes, and
amino precursor uptake and decarboxylation (APUD) cells. Neutrophils are also
typically present in smokers and patients with acute lung injury.
3. Pulmonary Circulation & Lymphatics
The lungs are supplied by two
circulations, pul-monary and bronchial. The bronchial circula-tion arises from
the left heart and sustains the metabolic needs of the tracheobronchial tree.
The bronchial circulation provides a small amount of blood flow (ie, less than
4% of the cardiac out-put). Branches of the bronchial artery supply the wall of
the bronchi and follow the airways as far as the terminal bronchioles. Along
their courses, the bronchial vessels anastomose with the pulmo-nary arterial
circulation and continue as far as the alveolar duct. Below that level, lung
tissue is sup-ported by a combination of the alveolar gas and pulmonary
circulation. Except for the main bron-chi within the mediastinum,
almost all the blood carried by the bronchial arteries enters the pulmo-nary
circulation.
The
pulmonary circulation normally receives the total output of the right heart via
the pulmonary artery, which divides into right and left branches to supply each
lung. Deoxygenated blood passes through the pulmonary capillaries, where O 2
is taken up and CO2 is eliminated. The oxygenated blood is then
returned to the left heart by four main pulmonary veins (two from each lung).
Although flows through the systemic and pulmonary circula-tions are equal, the lower pulmonary vascular
resis-tance results in pulmonary vascular pressures that are one-sixth of those
in the systemic circulation; as a result, both pulmonary arteries and veins
nor-mally have thinner walls than systemic vessels with less smooth muscle.
There are connections between the
bronchial and the pulmonary circulations. Direct pulmo-nary arteriovenous
communications, bypassing the pulmonary capillaries, are normally insignificant
but may become important in certain pathological states. The importance of the
bronchial circulation in contributing to the normal venous admixture is discussed
below.
Pulmonary Capillaries
Pulmonary capillaries are incorporated
into the walls of alveoli. The average diameter of these cap-illaries (about 10
µm) is barely enough to allow passage of a single red
cell. Because each capillary network supplies more than one alveolus, blood may
pass through several alveoli before reaching the pulmonary veins. Because of
the relatively low pressure in the pulmonary circulation, the amount of blood
flowing through a given capillary network is affected by both gravity and
alveolar size. Large alveoli have a smaller capillary cross-sectional area and
consequently increased resistance to blood flow. In the upright position,
apical capillaries tend to have reduced flows, whereas basal capillaries have
higher flows.
The pulmonary capillary endothelium has
rela-tively large junctions (5 nm wide), allowing the pas-sage of large
molecules such as albumin. As a result, pulmonary interstitial fluid is
relatively rich in albu-min. Circulating macrophages and neutrophils are able to
pass through the endothelium, as well as the smaller alveolar epithelial
junctions, with relative ease. Pulmonary macrophages are commonly seen in the
interstitial space and inside alveoli; they serve to prevent bacterial
infection and to scavenge for-eign particles.
Pulmonary Lymphatics
Lymphatic channels in the lung originate
in the interstitial spaces of large septa and are close to the bronchial
arteries. Bronchial lymphatics return flu-ids, lost proteins, and various cells
that have escaped in the peribronchovascular interstitium into the blood
circulation, thus ensuring homeostasis and permitting lung function. Because of
the large endo-thelial junctions, pulmonary lymph has a relatively high protein
content, and total pulmonary lymph flow may be as much as 20 mL/hr. Large
lymphatic vessels travel upward alongside the airways, form-ing the
tracheobronchial chain of lymph nodes. Lymphatic drainage channels from both
lungs com-municate along the trachea.
4. Innervation
The diaphragm is innervated by the phrenic
nerves, which arise from the C3–C5 nerve roots. Unilateral phrenic nerve block
or palsy only modestly reduces most indices of pulmonary function (about 25%)
in normal subjects. Although bilateral phrenic nerve palsies produce more
severe impairment, accessory muscle activity may maintain adequate ventilation
in some patients. Intercostal muscles are innervated by their respective
thoracic nerve roots. Cervical cord injuries above C5 are incompatible with
spon-taneous ventilation because both phrenic and inter-costal nerves are
affected.
The vagus nerves provide sensory
innervation to the tracheobronchial tree. Both sympathetic and parasympathetic
autonomic innervation of bron-chial smooth muscle and secretory glands is
pres-ent. Vagal activity mediates bronchoconstriction and increases bronchial
secretions via muscarinic receptors. Sympathetic activity (T1–T4) medi-ates
bronchodilation and also decreases secretions via β2-receptors.
Both α- and β-adrenergic receptors are present in the
pulmonary vasculature, but the sympathetic system normally has little effect on
pulmonary vascular tone. α1-Activity causes vasoconstriction; β2-activity mediates vasodilation.
Parasympatheticvasodilatory activity seems to be mediated via the release of
nitric oxide.
Related Topics