Chapter: Essential Microbiology: Microorganisms in Genetic Engineering
Expression vectors - Microorganisms in Genetic Engineering
Expression
vectors
Sometimes, the aim of a cloning experiment is not
just to obtain large amounts of a specific gene, but for the gene to be expressed. This involves using the host
cell as a sort of ‘factory’, to manufacture the specific protein encoded by the
cloned gene. One of the earliest applications of genetic engineering was the
production of human insulin in E. coli
(Figure 12.10). Insulin is needed in considerable quantities for the treat-ment
of diabetics; for years it was obtained from the pancreas of pigs and cattle,
but this had several disadvantages including immunological complications and
the risk of viral contamination. Insulin generated by recombinant means is free
from these prob-lems. Many proteins can now be produced in this way by
microorganisms at a rate several times that of the normal host cell. In order
for a gene to be expressed, it must have specific nucleotide sequences around it
that act as signals for the host cell’s tran-scription/translation machinery
(promoter, ribosomal binding site and terminator). Since these sequences differ
between, say, humans and E. coli, the
bacte-rial RNA polymerase will not recognise the human sequences, and
therefore, although a human gene may be cloned in E. coli using a simple vector, it will not be expressed. If,
however, the human gene could be inserted so that it was under the control of
the E. coli expression signals, then
transcription should take place. Specially designed vectors that provide these
signals are called expression vectors.
The choice of promoter sequence is particularly important; often, a strong
(i.e. very efficient) promoter is selected, so as to maximise the amount of
protein product obtained. Genes whose protein products are naturally
synthesised in abundance are likely to have such promoters. It is often helpful
to be able to regulate gene expression; inducible
promoters can be switched on and off by the presence of certain substances. The
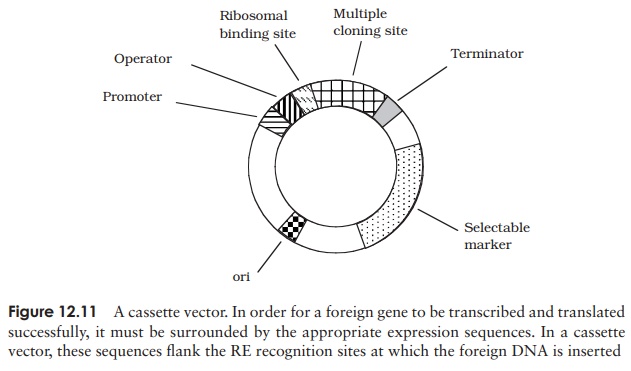
lac promoter (which controls the lacZ gene) is an example of this. Cassette vectors have promoter,
ribosomal binding site and terminator sequences clustered together as a
discrete unit, with a single recognition site for one or more REs being
situated downstream of the promoter (Figure 12.11).
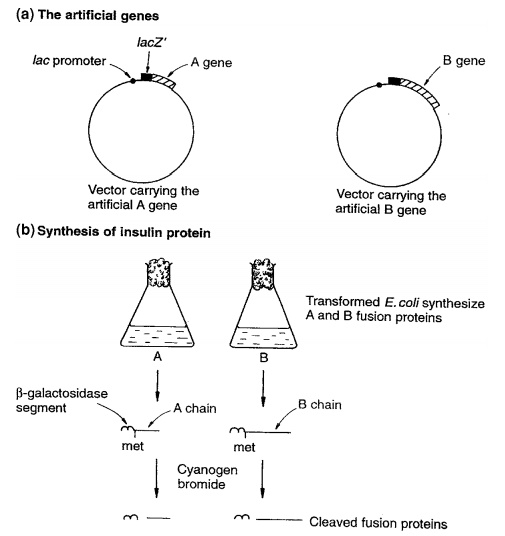

The small size of the insulin molecule (and gene) and
the size of the potential market made it a prime early candidate for production
by recombinant DNA technology. Most insulin used in the treatment of diabetes
nowadays is produced in this way. Systems based on E. coli have also been used to synthesise other small human
proteins with therapeutic potential such as human growth hormone,γ -interferon and tumour necrosis factor
(TNF).Bacteria, however, are not suitable host cells for theproduction of many
other human proteins such as tis-sue plasminogen activator (TPA) or blood
clotting Fac-tor VIII, due to the size and complexity of their genes. This is
because many proteins of complex eucaryotes are subject to post-translational modifications; this does not occur in
procaryotes, so bacteria such as E. coli
are not equipped with the cellular machinery to make the necessary
modifications to human proteins.
Another obstacle to the cloning of such proteins
concerns a fundamental difference in the way that procaryotic and eucaryotic
systems convert the message encoded in DNA to messenger RNA. Procaryotes lack
the means to remove introns, so if a human gene, for example, is expressed, the
whole of the primary transcript will be translated, instead of just the coding
sequences, leading to a non-functional protein. This problem can be
circumvented by cloning not the entire gene, but its cDNA, that is, just those
DNA sequences that are transcribed into mRNA and subse-quently translated into
amino acid sequences. This can be done by isolating mRNA, then using reverse
transcrip-tase to make a DNA copy. In the case of proteins such as insulin, the
very small size enabled artificial genes to be synthesised, based on their
known amino acid sequences.
Related Topics