Chapter: Mechanical : Engineering Thermodynamics : The Second Law of Thermodynamics
Equivalence of Kelvin-Planck and Clausius Statements
Equivalence of Kelvin-Planck
and Clausius Statements
The
Clausius and Kelvin-Planck statements of the second law are entirely
equivalent. This equivalence can be demonstrated by showing that the violation
of either statement can result in violation of the other one.
Referring to Figure
4.4(a) the device marked Clausius violator is pumping Q1 amount of
heat from a low temperature reservoir at T1 to a high temperature
reservoir at T2 without the aid of any external agency. This is an
impossible arrangement.
If
such an arrangement is possible it would also violate Kelvin-Planck statement.
Let a heat engine operating between the same reservoirs at T2 and T1
take in Q2 as heat input at T2. It converts a part of
this heat into work and rejects heat Q3to the sink at T1.
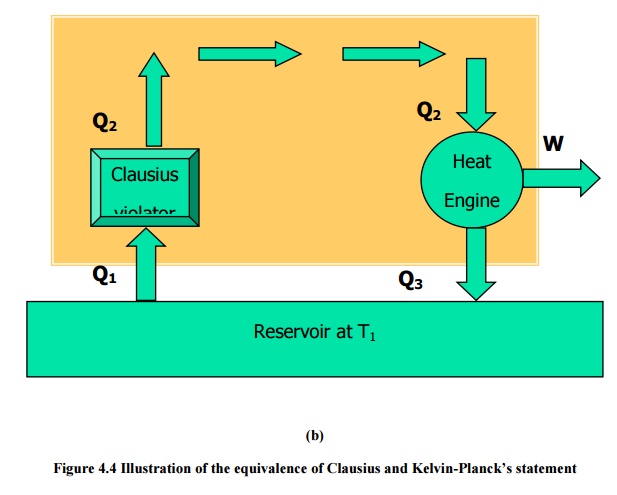
Since the Clausius violator is rejecting the same quantity Q2at T2,
it can be supplied directly into the heat engine so that the reservoir at T2
can be eliminated. This combination as shown in Figure 4.4 (b) is producing
continuous work with a single reservoir at T1. Hence it violates the
Kelvin-Planck statement.
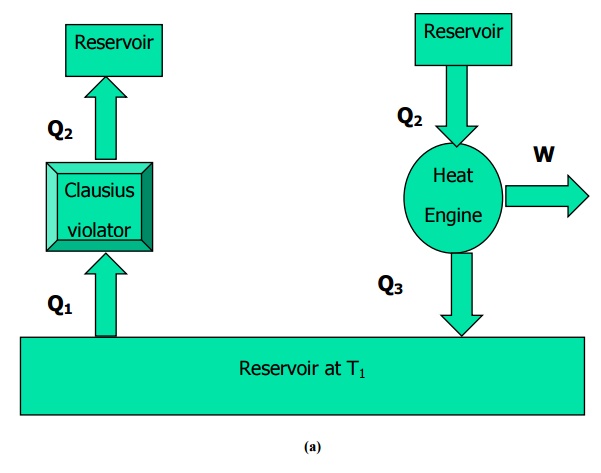
Figure
4.4 Illustration of the equivalence of Clausius and Kelvin-Planck’s statemen
Referring to Figure 4.5
a Kelvin-planck violator is converting all heat QH taken from the
reservoir at TH into work. If such an impossible heat engine is
assumed to exist it will violate the Clausius statement. Consider a
refrigerator pumping QL heat from the low temperature reservoir at TL
to the reservoir at higher temperature TH. Combined with the
Kelvin-Planck violator, the arrangement is pumping QL heat from TL
to TH, without any external agency. Hence it violate the Clausius
statement.
Related Topics