Chapter: Basic & Clinical Pharmacology : Antidepressant Agents
Drug Interactions - Clinical Pharmacology of Antidepressants
Drug Interactions
Antidepressants are
commonly prescribed with other psychotropic and nonpsychotropic agents. There
is potential for drug interac-tions with all antidepressants, but the most
serious of these involve the MAOIs and to a lesser extent the TCAs.
A. Selective Serotonin Reuptake Inhibitors
The most common
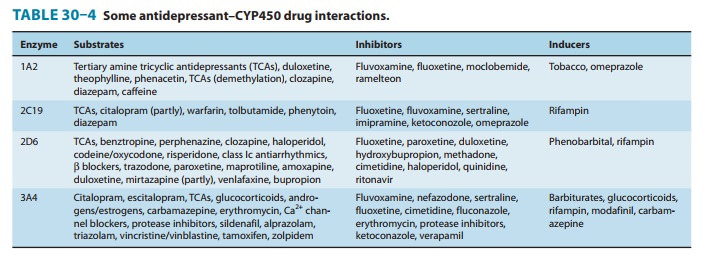
interactions with SSRIs are pharmacokinetic interactions. For example,
paroxetine and fluoxetine are potent CYP2D6 inhibitors (Table 30–4). Thus,
administration with 2D6 substrates such as TCAs can lead to dramatic and
sometimes unpredictable elevations in the tricyclic drug concentration. The
result may be toxicity from the TCA. Similarly, fluvoxamine, a CYP3A4
inhibitor, may elevate the levels of concurrently admin-istered substrates for
this enzyme such as diltiazem and induce bradycardia or hypotension. Other
SSRIs, such as citalopram and escitalopram, are relatively free of
pharmacokinetic interactions. The most serious interaction with the SSRIs are
pharmacody-namic interactions with MAOIs that produce a serotonin syn-drome .
B. Selective Serotonin-Norepinephrine Reuptake Inhibitors and Tricyclic Antidepressants
The SNRIs have
relatively fewer CYP450 interactions than the SSRIs. Venlafaxine is a substrate
but not an inhibitor of CYP2D6 or other isoenzymes, whereas desvenlafaxine is a
minor substrate for CYP3A4. Duloxetine is a moderate inhibitor of CYP2D6 and so
may elevate TCA and levels of other CYP2D6 substrates. Like all serotonergic
antidepressants, SNRIs are contraindicated in combination with MAOIs.
Elevations of TCA levels may occur when combined with CYP2D6 inhibitors or from constitutional factors. About 7% of the Caucasian population in the USA has a CYP2D6 polymorphism that is associated with slow metabolism of TCAs and other 2D6 substrates.
Combination
of a known CYP2D6 inhibitor and a TCA in a patient who is a slow metabolizer
may result in additive effects. Such an interaction has been implicated, though
rarely, in cases of TCA toxicity. There may also be additive TCA effects such
as anticholinergic or antihistamine effects when combined with other agents
that share these properties such as benztropine or diphenhydramine. Similarly,
antihypertensive drugs may exacerbate the orthostatic hypotension induced by
TCAs.
C. 5-HT2 Antagonists
Nefazodone is an
inhibitor of the CYP3A4 isoenzyme, so it can raise the level and thus
exacerbate adverse effects of many 3A4-dependent drugs. For example, triazolam
levels are increased by concurrent administration of nefazodone such that a
reduction in triazolam dosage by 75% is recommended. Likewise, administra-tion
of nefazodone with simvastatin has been associated with 20-fold increase in
plasma levels of simvastatin.
Trazodone is a
substrate but not a potent inhibitor of CYP3A4. As a result, combining
trazodone with potent inhibitors of CYP3A4, such as ritonavir or ketoconazole,
may lead to substan-tial increases in trazodone levels.
D. Tetracyclic and Unicyclic Antidepressants
Bupropion is
metabolized primarily by CYP2B6, and its metabolism may be altered by drugs
such as cyclophosphamide, which is a sub-strate of 2B6. The major metabolite of
bupropion, hydroxybupro-pion, is a moderate inhibitor of CYP2D6 and so can
raise desipramine levels. Bupropion should be avoided in patients taking MAOIs.
Mirtazapine is a
substrate for several CYP450 enzymes includ-ing 2D6, 3A4, and 1A2.
Consequently, drugs that inhibit these isozymes may raise mirtazapine levels.
However, mirtazapine is not an inhibitor of these enzymes. The sedating effects
of mir-tazapine may be additive with those of CNS depressants such as alcohol
and benzodiazepines.
Amoxapine and
maprotiline share most drug interactions com-mon to the TCA group. Both are
CYP2D6 substrates and shouldbe used with caution in combination with inhibitors
such as flu-oxetine. Amoxapine and maprotiline also both have anticholin-ergic
and antihistaminic properties that may be additive with drugs that share a
similar profile.
E. Monoamine Oxidase Inhibitors
MAOIs are associated
with two classes of serious drug interac-tions. The first of these is the
pharmacodynamic interaction of MAOIs with serotonergic agents including SSRIs,
SNRIs, and most TCAs along with some analgesic agents such as meperidine. These
combinations of an MAOI with a serotonergic agent may result in a
life-threatening serotonin syndrome
. The serotonin syndrome is thought to be caused by overstimula-tion of 5-HT
receptors in the central gray nuclei and the medulla. Symptoms range from mild
to lethal and include a triad of cogni-tive (delirium, coma), autonomic
(hypertension, tachycardia, diaphoreses), and somatic (myoclonus,
hyperreflexia, tremor) effects. Most serotonergic antidepressants should be
discontinued at least 2 weeks before starting an MAOI. Fluoxetine, because of
its long half-life, should be discontinued for 4–5 weeks before an MAOI is
initiated. Conversely, an MAOI must be discontinued for at least 2 weeks before
starting a serotonergic agent.
The second serious interaction with MAOIs occurs when an MAOI is combined with tyramine in the diet or with sympath-omimetic substrates of MAO. An MAOI prevents the breakdown of tyramine in the gut, and this results in high serum levels that enhance peripheral noradrenergic effects, including raising blood pressure dramatically. Patients on an MAOI who ingest large amounts of dietary tyramine may experience malignant hyperten-sion and subsequently a stroke or myocardial infarction. Thus, patients taking MAOIs require a low-tyramine diet and should avoid foods such as aged cheeses, tap beer, soy products, and dried sausages, which contain high amounts of tyramine . Similar sympathomimetics also may cause significant hyperten-sion when combined with MAOIs. Thus, over-the-counter cold preparations that contain pseudoephedrine and phenylpropa-nolamine are contraindicated in patients taking MAOIs.
Related Topics