Chapter: Basic & Clinical Pharmacology : Antidepressant Agents
Neurotrophic Hypothesis
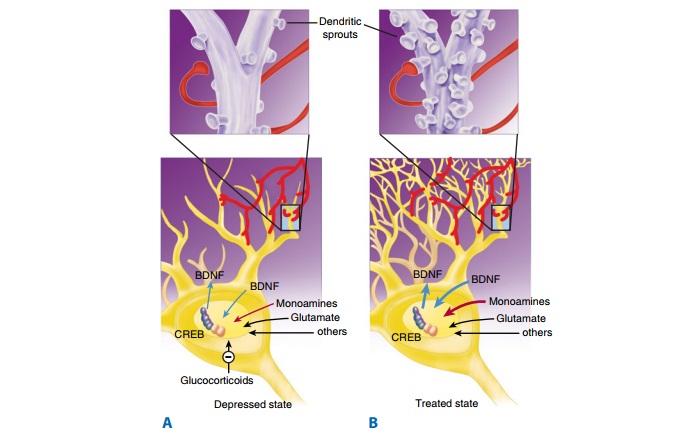
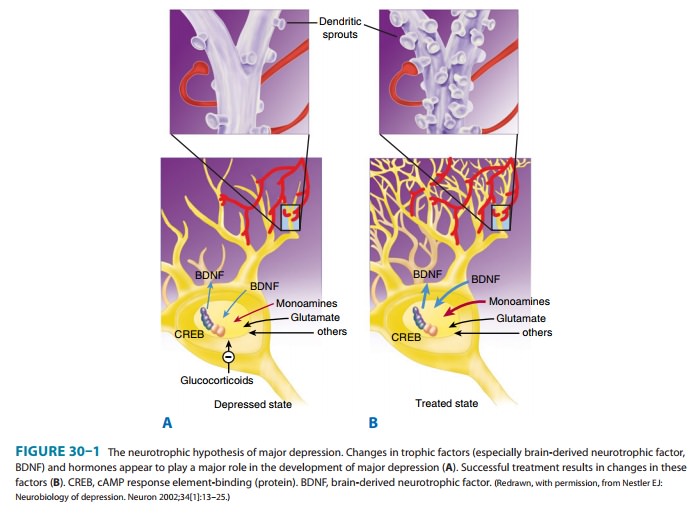
Neurotrophic Hypothesis
There is substantial evidence that nerve growth factors such as brain-derived neurotrophic factor (BDNF) are critical in theregulation of neural plasticity, resilience, and neurogenesis. The evidence suggests that depression is associated with the loss of neurotrophic support and that effective antidepressant therapies increase neurogenesis and synaptic connectivity in cortical areas such as the hippocampus. BDNF is thought to exert its influence on neuronal survival and growth effects by activating the tyrosine kinase receptor B in both neurons and glia (Figure 30–1).
Several lines of evidence support the neurotrophic hypothesis. Animal and human studies indicate that stress and pain are associ-ated with a drop in BDNF levels and that this loss of neurotrophic support contributes to atrophic structural changes in the hip-pocampus and perhaps other areas such as the medial frontal cortex and anterior cingulate.
The hippocampus is known to be important both in
contextual memory and regulation of the hypothalamic-pituitary-adrenal (HPA)
axis. Likewise, the anterior cingulate plays a role in the integration of
emotional stimuli and attention functions, whereas the medial orbital frontal
cortex is also thought to play a role in memory, learning, and emotion.
Over 30 structural
imaging studies suggest that major depres-sion is associated with a 5–10% loss
of volume in the hippocam-pus, although some studies have not replicated this
finding. Depression and chronic stress states have also been associated with a
substantial loss of volume in the anterior cingulate and medial orbital frontal
cortex. Loss of volume in structures such as the hip-pocampus also appears to
increase as a function of the duration of illness and the amount of time that
the depression remains untreated.
Another source of
evidence supporting the neurotrophic hypothesis of depression comes from
studies of the direct effects of BDNF on emotional regulation. Direct infusion
of BDNF into the midbrain, hippocampus, and lateral ventricles of rodents has
an antidepressant-like effect in animal models. Moreover, all known classes of
antidepressants are associated with an increase in BDNF levels in animal models
with chronic (but not acute) administration. This increase in BDNF levels is
consistently asso-ciated with increased neurogenesis in the hippocampus in
these animal models. Other interventions thought to be effective in the
treatment of major depression, including electroconvulsive ther-apy, also
appear to robustly stimulate BDNF levels and hippocam-pus neurogenesis in
animal models.
Human studies seem to
support the animal data on the role of neurotrophic factors in stress states.
Depression appears to be associated with a drop in BDNF levels in the
cerebrospinal fluid and serum as well as with a decrease in tyrosine kinase
receptor B activity. Conversely, administration of antidepressants increases
BDNF levels in clinical trials and may be associated with an increase in
hippocampus volume in some patients.
Much evidence supports
the neurotrophic hypothesis of depression, but not all evidence is consistent
with this concept. Animal studies in BDNF knockout mice have not always
sug-gested an increase in depressive or anxious behaviors that would be
expected with a deficiency of BDNF. In addition, some animal studies have found
an increase in BDNF levels after some types of social stress and an increase
rather than a decrease in depressive behaviors with lateral ventricle
injections of BDNF.
A proposed explanation
for the discrepant findings on the role of neurotrophic factors in depression
is that there are polymor-phisms for BDNF that may yield very different
effects. Mutations in the BDNF gene
have been found to be associated with altered anxiety and depressive behavior
in both animal and human studies.
Thus, the neurotrophic
hypothesis continues to be intensely investigated and has yielded new insights
and potential targets in the treatment of MDD.
Related Topics