Chapter: Pharmaceutical Biotechnology: Fundamentals and Applications : Production and Downstream Processing of Biotech Compounds
Cultivation Systems - Production of Biotech Compounds
Cultivation Systems
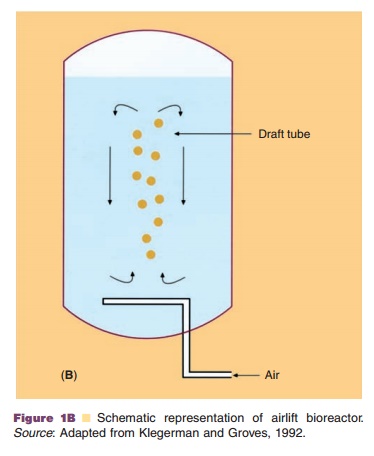
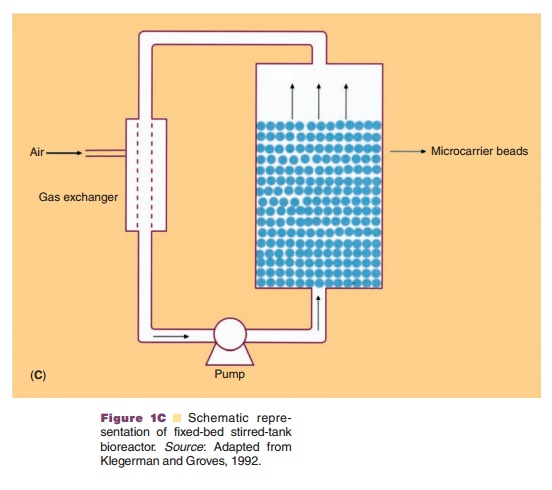
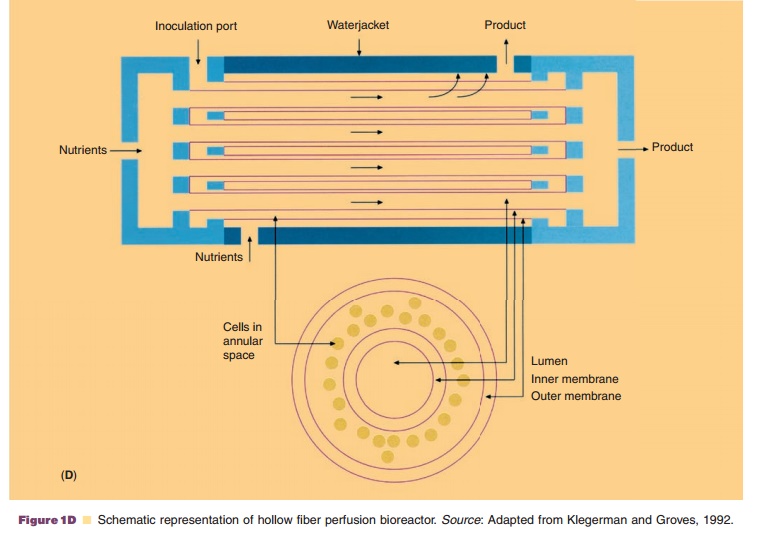
In general, cells can be cultivated in vessels containing an appropriate liquid growth medium in which the cells are either attached to microspheres, or free in suspension or in an immobilized state as monolayers, or entrapped in matrices (usually solidified with agar). The culture method will determine the scale of the separation and purification methods. Production-scale cultivation is commonly performed in fermentors or bioreactors. Bioreactor systems can be classified into four different types: stirred-tank, airlift, microcarrier (e.g., fixed-bed bioreactors) and membrane bioreactors (e.g., hollow fiber perfusion bioreactors) (Fig. 1). Because of its reliability and experience with the design and scaling up potential, the stirred tank is still the most commonly used bioreactor. The increased use of disposables in the manufacturing process has also resulted in the appearance of disposable bioreactors which take the form of large plastic bags which can be stirred or rocked to generate mixing for mass transfer. So far the application of this kind of technology for the manufacturing of commercial product has yet to be reported.
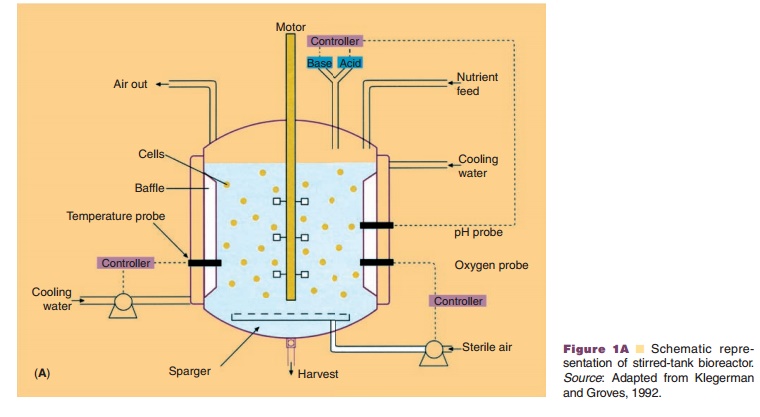
The kinetics of cell growth and product forma-tion will not only dictate the type of bioreactor used, but also how the growth process is run. Three types of fermentation protocols are commonly employed: (1) batch, (2) fed-batch, and (3) continuous production protocols. In all cases the cells go through four distinctive phases: lag, exponential growth, stationary, and death phase. The cell culture has to be free from undesired microorganisms that may destroy the cells or present hazards to the patient. This requires strict measures for both the procedures and materials used (Berthold and Walter, 1994; WHO 1998).
Examples of animal cells that are most com-monly used to produce proteins of clinical interest are Chinese hamster ovary cells (CHO), baby hamster kidney cells (BHK), lymphoblastoid tumor cells (interferon production), melanoma cells (plasminogen activator), and hybridized tumor cells (monoclonal antibodies).
Related Topics