Multiple choice questions - Choose the best answer: Basic concepts of organic reactions | 11th Chemistry : UNIT 12 : Basic concepts of organic reactions
Chapter: 11th Chemistry : UNIT 12 : Basic concepts of organic reactions
Choose the best answer: Basic concepts of organic reactions
Basic concepts of organic reactions | Chemistry
Evaluation
Choose the best answer
1. For the following reactions
(A) CH3CH2CH2Br + KOH → CH2 = CH2 + KBr +H2O
(B) (CH3)3CBr + KOH → (CH3)3 COH + KBr
(C) 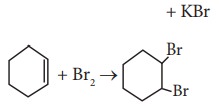
Which of the following statement is correct?
a) (A) is elimination, (B) and (C) are substitution
b) (A) is substitution, (B) and (C) are elimination
c) (A) and (B) are elimination and (C) is addition reaction
d) (A) is elimination, B is substitution and (C) is addition reaction.
2. What is the hybridisation state of benzyl carbonium ion?
(a) sp2
(b) spd2
(c) sp3
(d) sp2d
3. Decreasing order of nucleophilicity is
a) OH- > NH2- > -OCH3 > RNH2
b) NH2- > OH- > -OCH3 > RNH2
c) NH2- > CH3O- > OH- > RNH2
d) CH3O- > NH2- > OH- > RNH2
4. Which of the following species is not electrophilic in nature?
(a) Cl+
(b) BH3
(c) H3O+
(d) +NO2
5. Homolytic fission of covalent bond leads to the formation of
a) electrophile
b) nucleophile
c) Carbo cation
d) free radical
6. Hyper Conjugation is also known as
a) no bond resonance
b) Baker - nathan effect
c) both (a)and (b)
d) none of these
7. Which of the group has highest +I effect?
a) CH3-
b) CH3-CH2-
c) (CH3)2-CH-
d) (CH3)3-C-
8. Which of the following species does not exert a resonance effect?
a) C6H5OH
b) C6H5Cl
c) C6H5NH2
d) C6H5N+H3 or 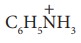
9. -I effect is shown by
a) -Cl
b) -Br
c) both (a) and (b)
d) -CH3
10. Which of the following carbocation will be most stable?
a) Ph3C-+
b) CH3-C(+)H2-
c) (CH3)2-C(+)H
d) CH2= CH – C(+)H2
11. Assertion: Tertiary Carbocations are generally formed more easily than primary Carbocations ions.
Reason: Hyper conjugation as well as inductive effect due to additional alkyl group stabilize tertiary carbonium ions.
a) both assertion and reason are true and reason is the correct explanation of assertion.
b) both assertion and reason are true but reason is not the correct explanation of assertion.
c) Assertion is true but reason is false
d) Both assertion and reason are false
12. Heterolytic fission of C-Br bond results in the formation of
a) free radical
b) Carbanion
c) Carbocation
d) Carbanion and Carbocation
13. Which of the following represent a set of nuclephiles?
a) BF3, H2O, NH2-
b) AlCl3, BF3, NH3
c) CN-, RCH2-, ROH
d) H+, RNH3+, :CCl2
14. Which of the following species does not acts as a nucleophile?
(a) ROH
(b) ROR
(c) PCl3
(d) BF3
15. The geometrical shape of carbocation is
(a) Linear
(b) tetrahedral
(c) Planar
(d) Pyramidal
Related Topics